Primary central nervous system lymphoma presenting with panhypopituitarism and diabetes insipidus
Montana Suwannasarn1, MD, Pongamorn Bunnag1, MD,
Wattana Mahattanakul2, MB, BS, MRCS,
Piyaphon Cheecharoen3, MD, Arunee Singhsnaeh4, MD.
1Department of Medicine,
2Department of Neurosurgery,
3Department of Radiology,
4Department of Pathology
Correspondence: Arunee Singhsnaeh, MD
Department of Pathology, Faculty of Medicine, Ramathibodi Hospital, Mahidol University,
Bangkok 10400, Thailand. Tel.: +66 2 201 1432
E-mail: arueed@gmail.com
Received: 20 February 2014; Accepted 10 March 2014
ABSTRACT
Lymphoma involving the hypothalamus is very rare. We present a case of a 48-year-old man with primary central nervous system lymphoma (PCNSL) presenting with hypothalamic and pituitary stalk lesions with frontal masses, causing diabetes insipidus and panhypopituitarism. Brain MRI with gadolinium enhancement demonstrated vivid enhancing hypothalamic and pituitary stalk lesions with frontal masses. Brain biopsy and subsequent histopathological examination were compatible with diffuse large B-cell non-Hodgkin’s lymphoma. After treatment with six cycles of intravenous high dose methotrexate-based chemotherapy, there was complete resolution of MRI finding. Steroid replacement was withdrawn but diabetes insipidus, hypothyroidism and hypogonadotrophic hypogonadism persisted. Regular follow up with adequate hormone replacement is crucial even there is apparent resolution of the tumor.
Introduction
Primary central nervous system lymphoma (PCNSL) is an uncommon form of non-Hodgkin’s lymphoma, comprising about 3% of all brain primary tumors that can affect any part of the brain or spinal cord1. Currently, PCNSL represents less than 1% of non-Hodgkin lymphoma2 mainly of the B-cell type and are most commonly associated with immunodeficiency. The incidence of PCNSL has increased only slightly in the past 10 years in individuals above the age of 60, and now stands at 0.5 per 100,000 patient-years.1
Isolated lesions are commonly seen in the frontal lobes, periventricular white matter, or the corpus callosum. PCNSL involving the hypothalamus, pituitary stalk, or pituitary gland with hypopituitarism in adult immunocompetent patients is very rare and only sporadic cases have been reported.3-6 In this article, we report a case of an immunocompetent PCNSL patient with frontal and hypothalamic lesions who presented with diabetes insipidus and anterior pituitary hormone deficiencies from lesions in the hypothalamus and pituitary stalk.
CASE HISTORY
A 48 -year-old man presented with a 2-month history of progressive headache and polyuria. He reported decreasing libido and decreased shaving for 4 months.
On physical examination, the patient was alert and cooperative. He had 20/70 and 20/50 visual acuity in the right and left eye respectively and his visual field was normal. Only a soft consistency of the bilateral testes was noted. His past medical history was unremarkable. He was found to have a serum sodium of 147 mmol/L, urine osmolality of 95 mosmol/kg. While serum osmolality was not collected, after dDAVP treatment, urine osmolality increased to 809 mosmol/kg. These findings were consistent with diabetes insipidus. Initial hormonal work up confirmed multiple anterior pituitary hormone deficiencies. The anterior pituitary axis hormones were decreased, except for prolactin (PRL) which was increased slightly at 48.29 ng/mL. Gonadotropin levels (FSH and LH) were low, while TSH level was normal. The morning cortisol level was low (Table 1). Magnetic resonance imaging (MRI) of the brain revealed an isointence T1 and hyperintense T2 mass involving the hypothalamus with homogeneous and vivid enhancement, exerting pressure effect to the optic chiasm. The pituitary stalk was displaced downward with some thickening at the proximal part of the pituitary stalk. There were also two masses in the right frontal region with massive vasogenic edema. The lesions were isointense on T1-weighted images with homogenously vivid enhancement after gadolinium administration. The pituitary gland architecture was well preserved (Figure 1).

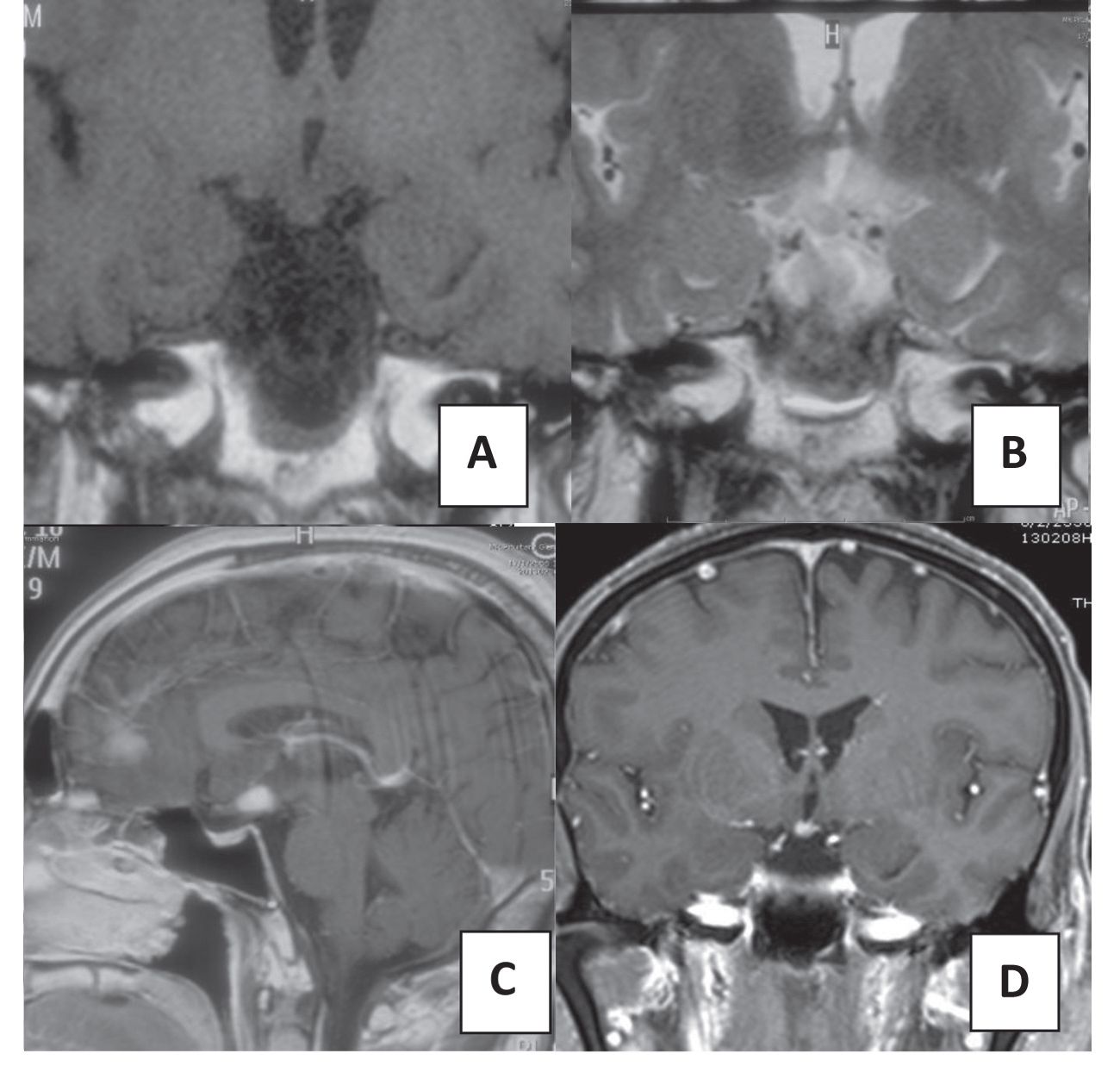
Figure 1 A, B: There is an isointense T1 and hyperintense T2mass involving the hypothalamus with pressure effect to the optic chiasm. C,D: On post Gadolinium T1 weighted images, the lesions show homogeneous and vivid enhancement
Due to massive vasogenic edema and adrenal insufficiency, high dose dexamethasone was given. Then the patient underwent an endoscopic tumor biopsy and a craniotomy with tumor removal from frontal lobe lesion. Desmopressin (dDAVP) and pituitary hormone replacement with glucocorticoids and levothyroxine were given. His general condition, polyuria and electrolytes abnormalities improved markedly postoperatively.
Histopathological examination of the surgical specimen showed neoplastic cells with abundant cytoplasm and pleomorphic nuclei with considerable mitotic activity (Figure 2). These atypical large cells were positive for the leukocyte common antigen (LCA). Malignant lymphoma was diagnosed and these cells were diffusely positive for CD20 which indicated B-cell origin. In primary CNS lymphoma, more than 95% are diffuse large B-cell lymphoma, the others are low-grade B-cell lymphoma, other types of B-cell lymphoma such as Burkitt lymphoma, T-cell lymphoma and Hodgkin lymphoma7. Beside CD20. The tumor cells were positive for Bcl-6, MUM1, focally positive 20% for Bcl-2 and Ki67 positive 90% of tumor nuclei. While the specific T-lymphocyte marker, CD3 was negative and CD5,CD10 CyclinD1 and EBER were negative all7. The histopathological and immunohistochem-istry results were compatible with the diagnosis of a diffuse large B-cell non-Hodgkin’s lymphoma. Computed tomography (CT) of chest and abdomen (including bone marrow examination) excluded other neoplastic foci. HIV testing was negative. The patient was treated with 6 cycles of high dose intravenous methotrexate (3.5 mg/1.73 m2 ) and Ara-C.
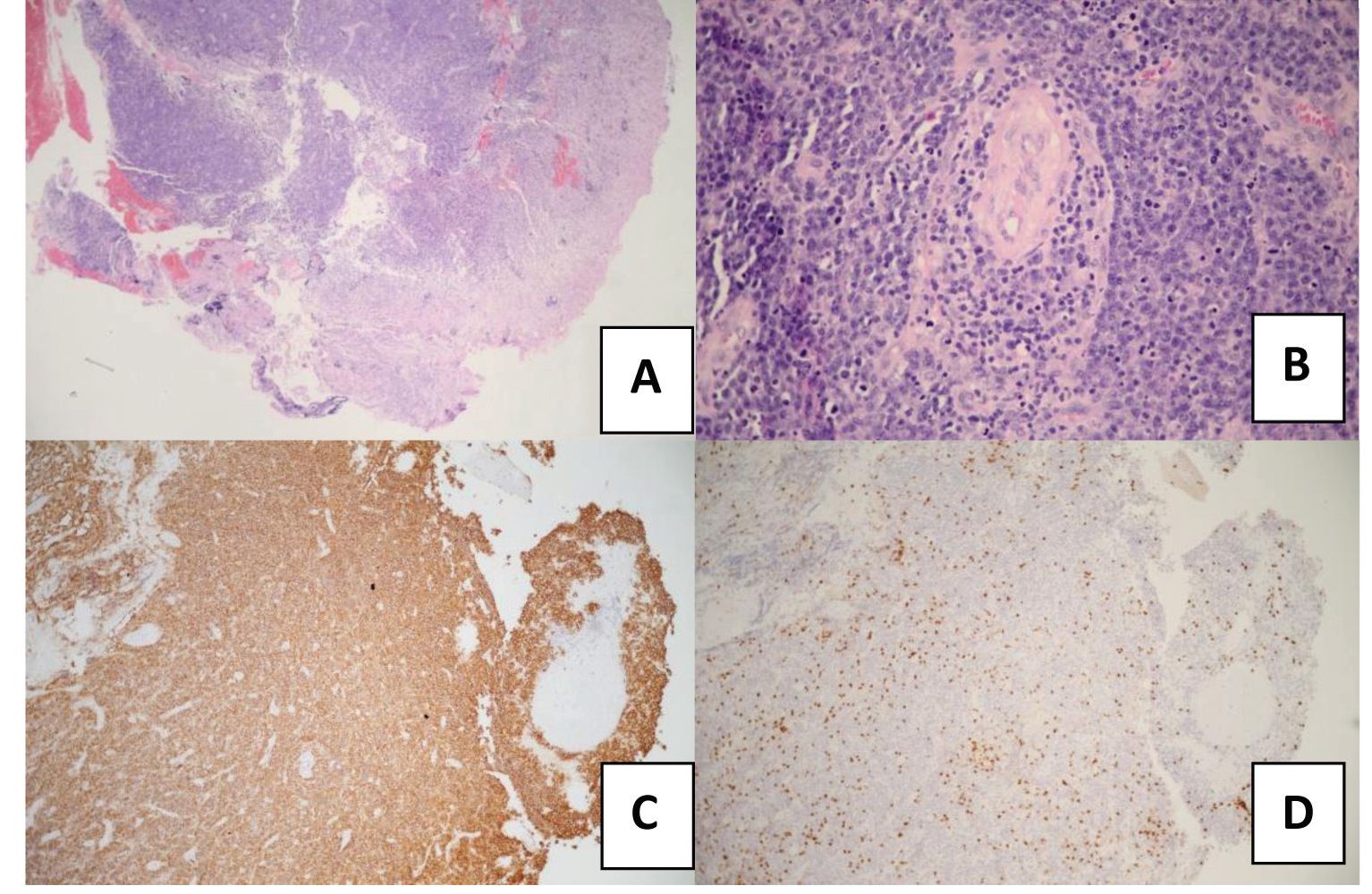
Figure 2 Tumor histopathology. A : Tumor cell in infiltration pattern (40X), B : Atypical large tumor cells infiltration in angiocentric pattern (400X), C: Membrane staining of the tumor cells with an antibody to CD20, indicating a B-cell origin(100X) D: Negative membrane staining for CD3(100X)
At his 6-month follow-up, complete radiological remission of the lesions was achieved with recovery of secondary adrenal insufficiency. Steroid replacement was then withdrawn. However, his diabetes insipidus and secondary hypothyroidism persisted, necessitating thyroid hormone, testosterone and desmopressin therapy.
DISCUSSION
Primary central nervous system lymphoma (PCNSL) with hypothalamic involvement presenting with DI and hypopituitarism has been reported in few patients. Although CNS lymphoma occurs more frequently in immunocompromised patients, incidence peaked in the mid-1990s and has now declined. This might be related to changes in HIV incidence and management 2 8. In contrast, the incidence remains high among older patients (>60 years) who are mostly immunocompetent9. The reason for this increasing incidence is unknown.
Unlike our case, most of the previously cases had isolated hypothalamic or pituitary involvement, making the diagnosis of lymphoma more difficult. Our patient was an immunocompetent patient presenting with frontal lesions together with diabetes insipidus, hypopituitarism and hyperprolactinemia resulting from hypothalamic involvement. The MRI findings included homogenously enhancing masses in the hypothalamus and frontal area, which are typical for PCNSL. Histopathological examination confirmed the diagnosis of PCNSL at both lesions.
Although the prognosis remains poor for the majority of patients, approximately 20-30% of cases can be cured2. The best treatment strategy has yet to be defined10, 11. However, biopsy followed by corticosteroids, high dose methotrexate (MTX)-based chemotherapy and/or radiotherapy are the current treatment options. PCNSL is a highly radiosensitive and chemosensitive tumor2 which can result in a resolution of the tumors, however anterior pituitary hormone deficiency and diabetes insipidus generally persist. Due to irreversible damage of the hypothalamus, regular follow up and adequate hormone replacement therapy are crucial even there is apparent resolution of the tumor.
REFERENCE
1. Hochberg FH, Baehring JM, Hochberg EP. Primary CNS lymphoma. Nat Clin Pract Neurol. 2007 Jan;3(1):24-35.
2. Sierra del Rio M, Rousseau A, Soussain C, Ricard D, Hoang-Xuan K. Primary CNS lymphoma in immunocompetent patients. Oncologist. 2009 May;14(5):526-39.
3. Layden BT, Dubner S, Toft DJ, Kopp P, Grimm S, Molitch ME. Primary CNS lymphoma with bilateral symmetric hypothalamic lesions presenting with panhypopituitarism and diabetes insipidus. Pituitary. 2011 Jun;14(2):194-7.
4. Katz BJ, Jones RE, Digre KB, Warner JE, Moore KR. Panhypopituitarism as an initial manifestation of primary central nervous system non-Hodgkin’s lymphoma. Endocr Pract. 2003 Jul-Aug;9(4):296-300.
5. Malaise O, Frusch N, Beck E, Servais S, Caers J, de Prijck B, et al. Panhypopituitarism and diabetes insipidus in a patient with primary central nervous system lymphoma. Leuk Lymphoma. 2012 Dec;53(12):2515-6.
6. Capra M, Wherrett D, Weitzman S, Dirks P, Hawkins C, Bouffet E. Pituitary stalk thickening and primary central nervous system lymphoma. J Neurooncol. 2004 Mar-Apr;67(1-2):227-31.
7. Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, et al. The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol. 2007 Aug;114(2): 97-109.
8. Hoffman S, Propp JM, McCarthy BJ. Temporal trends in incidence of primary brain tumors in the United States, 1985-1999. Neuro Oncol. 2006 Jan;8(1):27-37.
9. Kadan-Lottick NS, Skluzacek MC, Gurney JG. Decreasing incidence rates of primary central nervous system lymphoma. Cancer. 2002 Jul 1;95(1):193-202.
10. O’Neill BP, O’Fallon JR, Earle JD, Colgan JP, Brown LD, Krigel RL. Primary central nervous system non-Hodgkin’s lymphoma: survival advantages with combined initial therapy? Int J Radiat Oncol Biol Phys. 1995 Oct 15;33(3):663-73.
11. Giustina A, Gola M, Doga M, Rosei EA. Clinical review 136: Primary lymphoma of the pituitary: an emerging clinical entity. J Clin Endocrinol Metab. 2001 Oct;86(10):4567-75.


