Primary Testicular NK/T-cell Lymphoma presented as testicular abscess: A Case Report and Literature Review of 33 cases
Sanya Sukpanichnant • Yingyong Chinthammitr • Naravat Poungvarin
Department of Pathology, Faculty of Medicine Siriraj Hospital, Mahidol University, Bangkok 10700, Thailand
Corresponding author: Sanya Sukpanichnant, M.D.,
Department of Pathology, Faculty of Medicine Siriraj Hospital, Mahidol University, Bangkok 10700, Thailand,
Tel/Fax +66 2 411 4260 e-mail: sanya.suk@mahidol.ac.th
Coauthors and names of the department and institution:
Yingyong Chinthammitr, M.D.
Department of Medicine, Faculty of Medicine Siriraj Hospital, Mahidol University, Bangkok, Thailand
Naravat Poungvarin, M.D., Ph.D.
Department of Clinical Pathology, Faculty of Medicine Siriraj Hospital, Mahidol University, Bangkok, Thailand
Abstract
Primary testicular NK/T-cell lymphoma is rare. We report a 61-year-old Thai man who presented with right testicular abscess clinically but tissue obtained from incision and drainage was proven to be NK/T-cell lymphoma. The lymphoma cells were small to medium in size and showed angiocentricity with associated necrosis. The lymphoma cells were CD3+ (cytoplasmic), CD43+, CD56+, TIA-1+, Ki-67+ (nearly 100%) and EBV encoded RNA (EBER)+. Clinical staging was IE. CHOP chemotherapy and intrathecal methotrexate for primary CNS prophylaxis were administered with partial reduction of the testicular mass. However, 5.5 months after diagnosis, the patient developed left testicular enlargement and left facial palsy caused by leptome-ningeal involvement. The patient died of progressive disease 9 months after diagnosis. The clinical course in this patient was aggressive, similar to those described in the literature review except for the uncommon CNS involvement. Based on 19 cases of primary testicular lymphoma (including the present case) and 15 cases of testicular involvement by NK/T-cell lymphoma elsewhere, the former seem to be less aggressive than the latter but the difference is not statistically significant in either overall median survival (32 vs. 16 weeks, p = 0.086) or mean survival (29.4 vs. 17.9 weeks, p = 0.083).
Keywords: Testicular lymphoma • testicular abscess • extranodal • NK/T-cell lymphoma • EBER • CNS involvement
INTRODUCTION
Testicular lymphoma is uncommon, accounting for only 1% of extranodal lymphoma in Thailand [1]. At Siriraj Hospital, more than 80% of testicular lymphoma cases were diffuse large B-cell lymphoma while only 9% were mature T-cell lymphoma. Only a small number of testicular involvement by nasal-type extranodal NK/T-cell lymphoma have been reported in the English literature. No primary testicular lymphoma case was included in the study of 67 cases of extranodal NK/T-cell lymphoma previously reported from Thailand [2]. In the English literature, primary testicular lymphoma of any subtypes is uncommon and it mostly presents as stage I or II disease (approximately 75% of the patients). However, it has been well known for aggressiveness in term of systemic disease at relapse (30-40% of cases). CNS involvement, in the form of brain parenchyma, leptomeninges or both, is the most common site at relapse in nearly half of the patients [3-5]. We hereby report a case of primary testicular NK/T-cell lymphoma who first presented as testicular abscess and review the English literature to emphasize the clinical importance of this particular type of lymphoma.
CLINICAL HISTORY
A previously healthy, 61-year-old, Thai man had presented with a right testicular mass for 4 months and later he experienced testicular pain and fever. He was diagnosed as having testicular abscess at a local hospital. Incision and drainage (I&D) was performed and oral antibiotics were given. The tissue with some necrotic debris was obtained and submitted for microbiological study and pathologic examination. The microbiological study yielded no growth of microorganism but the pathologic diagnosis given by a pathologist at a private pathology laboratory based on histologic findings only was malignant round cell tumor, suspicious of malignant lymphoma. Immunohisto-chemistry was recommended for further evaluation. The patient was then referred to Siriraj Hospital for further management.
Three weeks after I&D, the patient attended the outpatient department service of the Division of Urology, Department of Surgery at Siriraj Hospital. No other significant clinical history was obtained except for night sweats. Physical examination revealed an enlarged right testis, measuring 15 cm. in maximal diameter. The small healed I&D wound was noted. No lymphadenopathy or organomegaly was detected. Complete blood count and blood chemistry results were within the normal ranges. The histologic sections and corresponding tissue blocks were submitted to the Department of Pathology for review. The diagnosis was extranodal NK/T-cell lymphoma, nasal type, according to the WHO classification (2008) [6].
Two weeks after the first visit at Siriraj Hospital, the patient underwent bone marrow examination and computed tomography (CT) of the chest and whole abdomen for clinical staging which corresponded to stage IE. Cerebrospinal fluid (CSF) was also obtained and no lymphoma cells were detected. The International Prognostic Index (IPI) was low (IPI = 1). So, 10 days after a definite diagnosis or 37 days after I&D, the patient received CHOP regimen and intrathecal methotrexate administration for primary CNS prophylaxis. The patient refused both orchiectomy and local radiotherapy. A week later, the right testicular mass reduced in size to 9x6 cm. Two weeks later, the ear, nose and throat (ENT) region was examined at the Department of Otolaryngology. No abnormalities were detected. Partial response to CHOP chemotherapy was obtained after the fourth course of CHOP as the right testicular mass reduced in size to 5.2 cm.
in maximal diameter. But, after the fifth course of chemotherapy, the right testicular mass grew up to 7 cm. and, 3 weeks after the seventh course of chemotherapy or 5.5 months after diagnosis, the patient developed generalized bone pain and additional left testicular enlargement to 10 cm. in maximal diameter. Oral prednisolone (20 mg./day) was given as a palliative treatment. Three days later, the patient developed left facial palsy caused by leptomenin-geal involvement as lymphoma cells were detected in the CSF. He received palliative oral chemotherapy and further intrathecal therapy (hydrocortisone, methotrexate, and Ara-C) twice weekly without any improvement. A week later, CT of the brain revealed a subacute lacunar infarction at left basal ganglion and enhancing soft tissue lesions at both nasal cavities and paranasal sinuses. The patient also developed pain at both legs that progressed to be cauda equina syndrome, requiring urinary bladder catheter dwelling. During admission and while receiving palliative treatment, the patient developed febrile neutropenia and influenza A pneumonia but he recovered eventually from pneumonia. During this time, the right and left testes measured 6 cm. and 10 cm. in maximal diameter respectively. Local irradiation was given at both testes for a 10 day period and discontinued as the patient asked for only palliative care. He was discharged 7.5 months after diagnosis and received palliative medication including oral prednisolone (20 mg./day), morphine syrup and antipyretics. During this terminal phase, the patient had suffered from progressive fatigue, muscle pain, bone pain, anorexia and fever. He died of progressive disease 9 months after diagnosis at a local hospital.
MATERIALS AND METHODS
The provided paraffin-embedded formalin-fixed tissue block of the tissue obtained from I&D was used for histopathologic, histochemical, im-munohistochemical, in situ hybridization (ISH) and molecular genetic studies. Standard hematoxylin & eosin-stained slide and PAS-stained slide were performed conventionally. Immunohistochemical staining and ISH for EBV-encoded RNA (EBER) were performed by Bench-Mark® XT autostainer (Roche Diagnostic). The authors could usefully provide more detail on antibody clones in dilutions in the Methods section. The remaining tissue in the block was used for molecular genetic studies for rearrangement of T-cell receptor (TCR) genes using BIOMED-2 multiplex PCR-based assay kit. The cytologic preparation was prepared from the provided CSF using cytospin technique.
RESULTS
The malignant round cells described in the original pathology report appeared to be small to medium-sized lymphoma cells, showing diffuse infiltration with angiocentricity and multifocal necrosis. Immunohistochemistry demonstrated that the lymphoma cells were CD3+ (cytoplasmic), CD43+, CD56+, TIA-1+ and Ki-67+ (nearly 100%). Other markers were negative including betaF-1, CD4, CD5, CD8, CD20, CD34, CD68, CD117, myeloperoxidase and TdT. Diffuse and strong positivity with in-situ hybridization for EBV RNA (EBER) was noted in the lymphoma cells. (Figure 1) Molecular genetic study for rearrangement of TCR genes was performed but the target fragment was not successfully amplified due to insufficient DNA quality. Finally, the diagnosis of extranodal NK/T-cell lymphoma, nasal type, was given according to the WHO classification (2008) [6]. Lymphoma cells were found in the CSF at the time of left facial palsy, indicating leptomeningeal involvement. Only few cells had few azurophilic granules. (Figure 2)
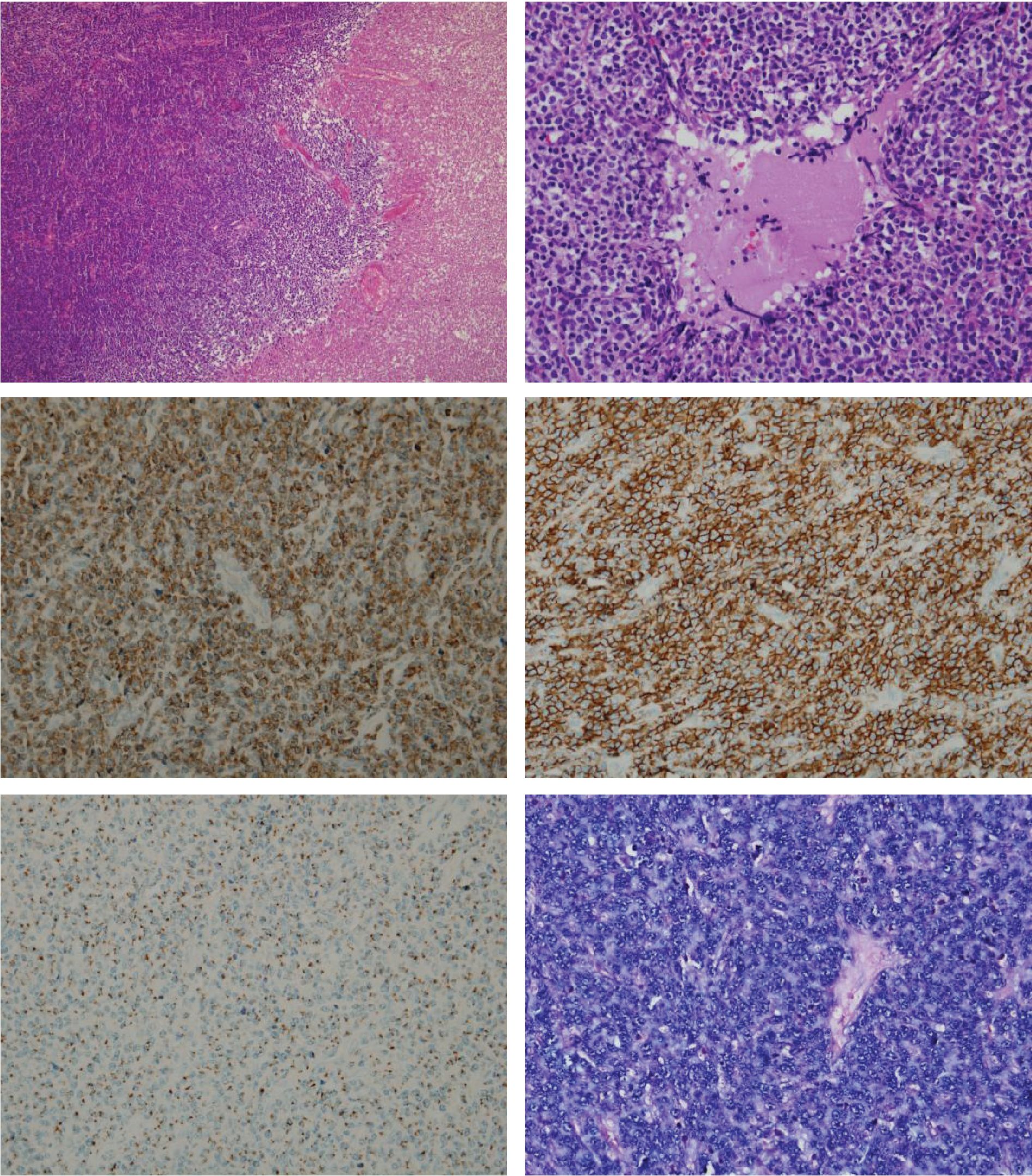
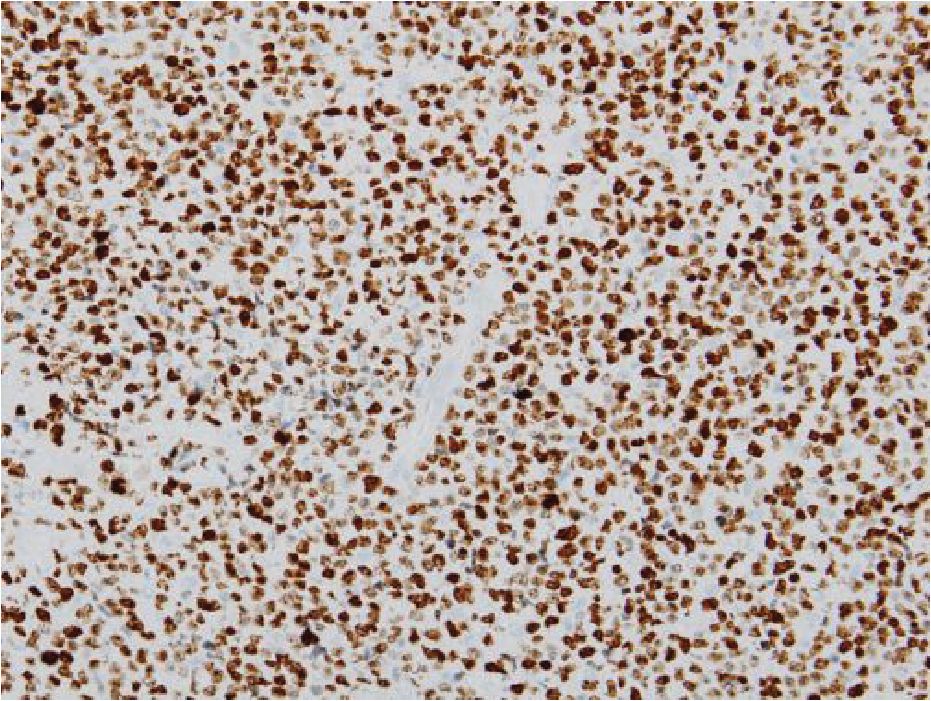
Figure 1 Primary testicular extranodal NK/T-cell lymphoma, nasal type: a Necrosis at one part of the lesion (H&E); b Small to medium-sized lymphoma cells with angiocentricity (H&E); c CD3+, cytoplasmic pattern (anti-CD3); d CD56+ (anti-CD56); e TIA-1+ (anti-TIA-1); f EBER+ (ISH method); and g Ki-67+ (anti-MIB-1).
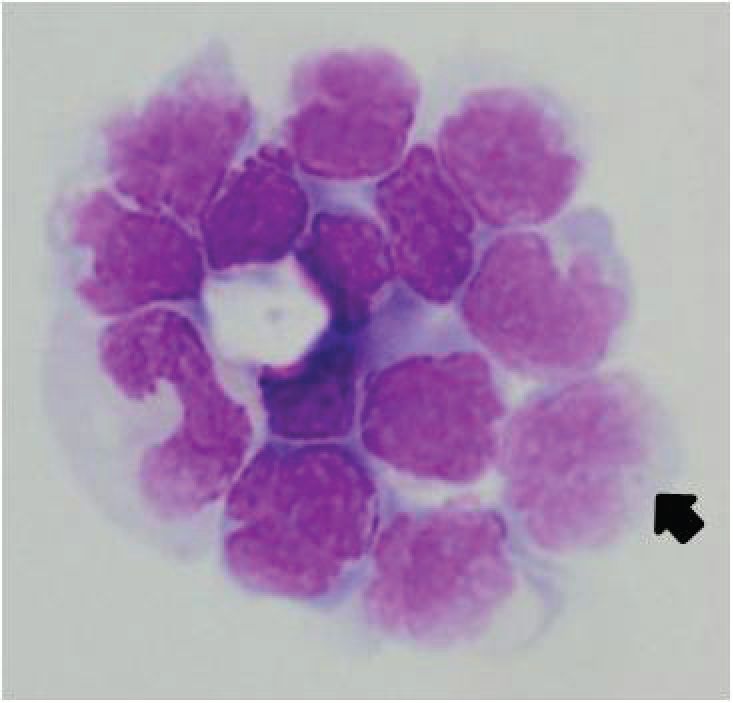
Figure 2 Lymphoma cells in the cerebrospinal fluid. Note the pleomorphic nuclei but few azulophilic granules (arrow).
DISCUSSION
The diagnosis of nasal-type NK/T-cell lymphoma requires the conventional approach according to the WHO classification (2008) [6]. After clinical staging work-up, the lesion was localized to the testis and the morphologic, immunopheno-typic and genetic findings confirmed that this is primary testicular NK/T-cell Lymphoma. Azurophilic granules recognized in Wright or Diff-Quik stained cytologic preparation will be very helpful to indicate NK-cell or cytotoxic T-cell phenotype of the lymphoma cells despite the nuance of cytologic features including lymphoblastic, small, small to medium, medium, medium to large and large pleomorphic or anaplastic cells. Syncytial aggregates or small clusters of large pleomorphic cells can be seen in fine needle aspiration [7]. It is noteworthy to be aware that azurophilic granules may be found in only some of the lymphoma cells [8]. In this present case, azurophilic granules were hard to find in the CSF. In the histologic sections, the tissue necrosis caused by lymphoma cells infiltrating around the blood vessel (angiocentricity) as well as angiode-struction is a salient feature described in this particular type of lymphoma [6, 8]. But in some case reports, only coagulation necrosis and ulceration were described [9]. Softening of tissue following necrosis occurred in the testicular mass then led to the clinical impression of testicular abscess in the setting of a testicular mass with pain and fever as diagnosed at a local hospital in the present case. Thus, testicular lymphoma should be kept in mind when encountered with testicular abscess. Tissue obtained from incision and drainage should be submitted not only microbiological study but also pathologic examination.
The most common type of primary testicular lymphoma is diffuse large B-cell lymphoma (more than 90% of cases in most series). The less common types include extranodal NK/T-cell lymphoma and Burkitt lymphoma [3, 10]. CNS involvement with intracerebral, leptomeningeal or both involvements has been noted in up to 30% of patients with testicular B cell lymphoma [11]. Possibly similar to other testicular tumors, CNS metastasis in malignant lymphoma can occur through lymphatic drainage to the retroperitoneal lymph nodes, then to the posterior mediastinal or supraclavicular lymph nodes before spreading through the systemic circulation. Direct hematogenous spread to the brain is also possible [12]. Blood-brain barrier (BBB) provides a sanctuary hiding place for lymphoma cells that spread to the CNS as most standard chemotherapeutic agents cannot penetrate across the BBB. So CNS becomes a common site of relapse after standard chemotherapy regimens. Prophylactic intrathecal chemotherapy or systemic chemotherapeutic agents that can penetrate across the BBB, including methotrexate and cytarabine, have been recommended to prevent leptomeningeal or CNS involvement [3, 5]. Prophylactic intrathecal chemotherapy alone, however, may not be able to prevent brain parenchymal involvement at relapse; thus whole brain radiotherapy or systemic chemotherapy are needed [3]. While CNS involvement is quite common in primary testicular large B-cell lymphoma, CNS involvement in primary testicular NK/T-cell lymphoma was reported in only 1 case in the literature proven by Kim and colleague [13] and in the present case. There were 2 other cases in the literature mentioned about encephalitis [14] and intracranial bleeding [15] but they did not confirm whether there was genuine CNS involvement or not. At this point, only 2 out of 19 cases of primary testicular NK/T-cell lymphoma in the literature (including the present case) had CNS involvement (10.5%).
From the compilation of three large series of 98 cases of nasal-type extranodal NK/T-cell lymphoma (non-nasal site) from Hong Kong, Singapore and Korea, only 9 cases had testicular involvement (9.2%) or approximately 5.5% of all nasal-type extranodal NK/T-cell lymphoma of both nasal and non-nasal sites. None of these 9 cases had CNS involvement. All patients died of disease shortly, varying from 1 to 40 weeks. The patients usually showed a good initial response to chemotherapy but commonly relapsed [8, 16, 17]. In contrast to the pattern of dissemination of diffuse large B-cell lymphoma of the testis, regional lymph node involvement is quite uncommon in primary testicular NK/T cell lymphoma, even with widely disseminated disease [18].
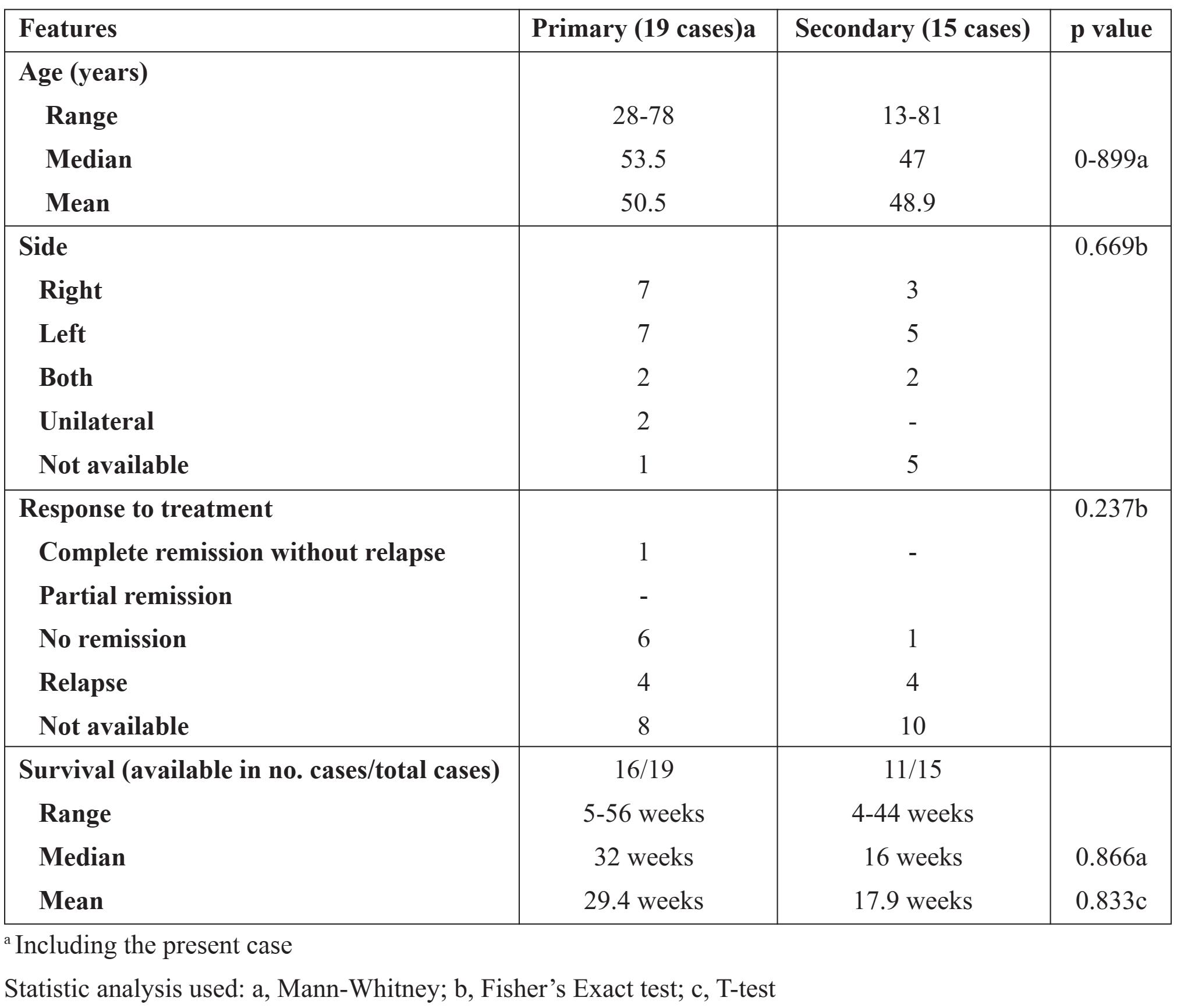
In the literature review of nasal-type NK/T-cell lymphoma with testicular involvement (Table 1), there are 19 cases of primary testicular lymphoma (including the present case) and 15 cases of testicular involvement as secondary lymphoma. Only one case report by Guler et al. [20] had a presumptive diagnosis of abscess or malignancy. We regarded a 5 5-year-old patient reported by Chan et al. [18] as primary testicular lymphoma, in contrast to the literature review by Liang et al. [27] that placed him as non-primary testicular case, because this particular case presented first with left testicular mass then 1 month after orchiectomy he developed cutaneous involvement without bone marrow or cerebrospinal fluid involvement. After initiation of combination chemotherapy, nasopharyngeal examination revealed lymphomatous involvement. We also included the two cases of primary testicular lymphoma reported by Lin et al. [9] in this literature review. The comparison between the primary testicular and secondary testicular groups is shown in Table 2. The median age of patients with primary testicular NK/T-cell lymphoma was not different from that of patients with secondary lymphoma (53.5 vs. 47, p = 0.899) while the mean age of the former is only 1.6 years older than the latter (50.5 vs. 48.9). The primary testicular NK/T-cell lymphoma group seems to behave less aggressively than the secondary testicular lymphoma group but there is not statistically significant difference in either overall median survival (32 vs. 16 weeks, p = 0.086) or mean survival (29.4 vs. 17.9 weeks, p = 0.083). In some cases of primary testicular NK/T-cell lymphoma reported in the literature, the disease progressed very rapidly. Involvement of other sites was documented within a month after orchiectomy.
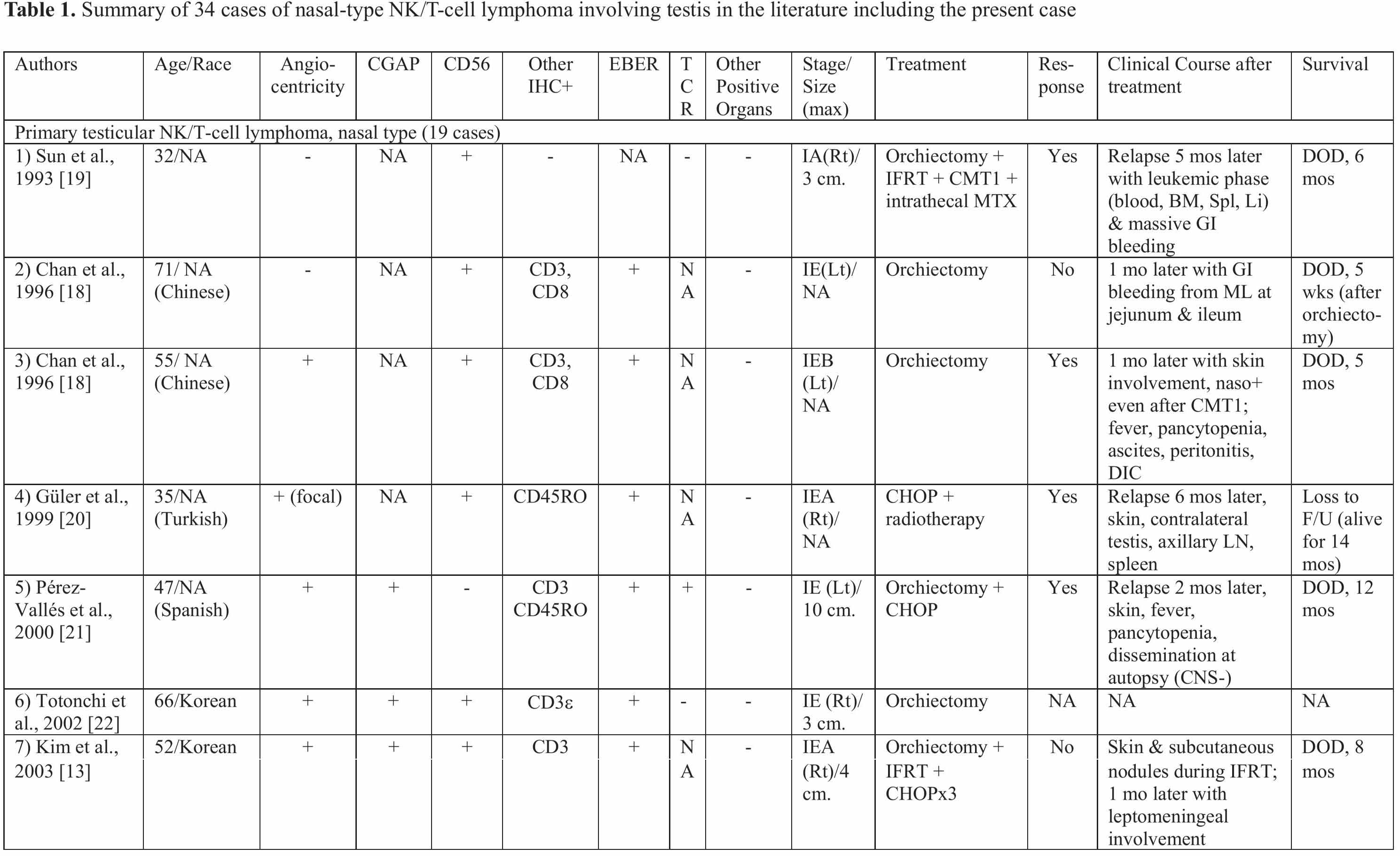
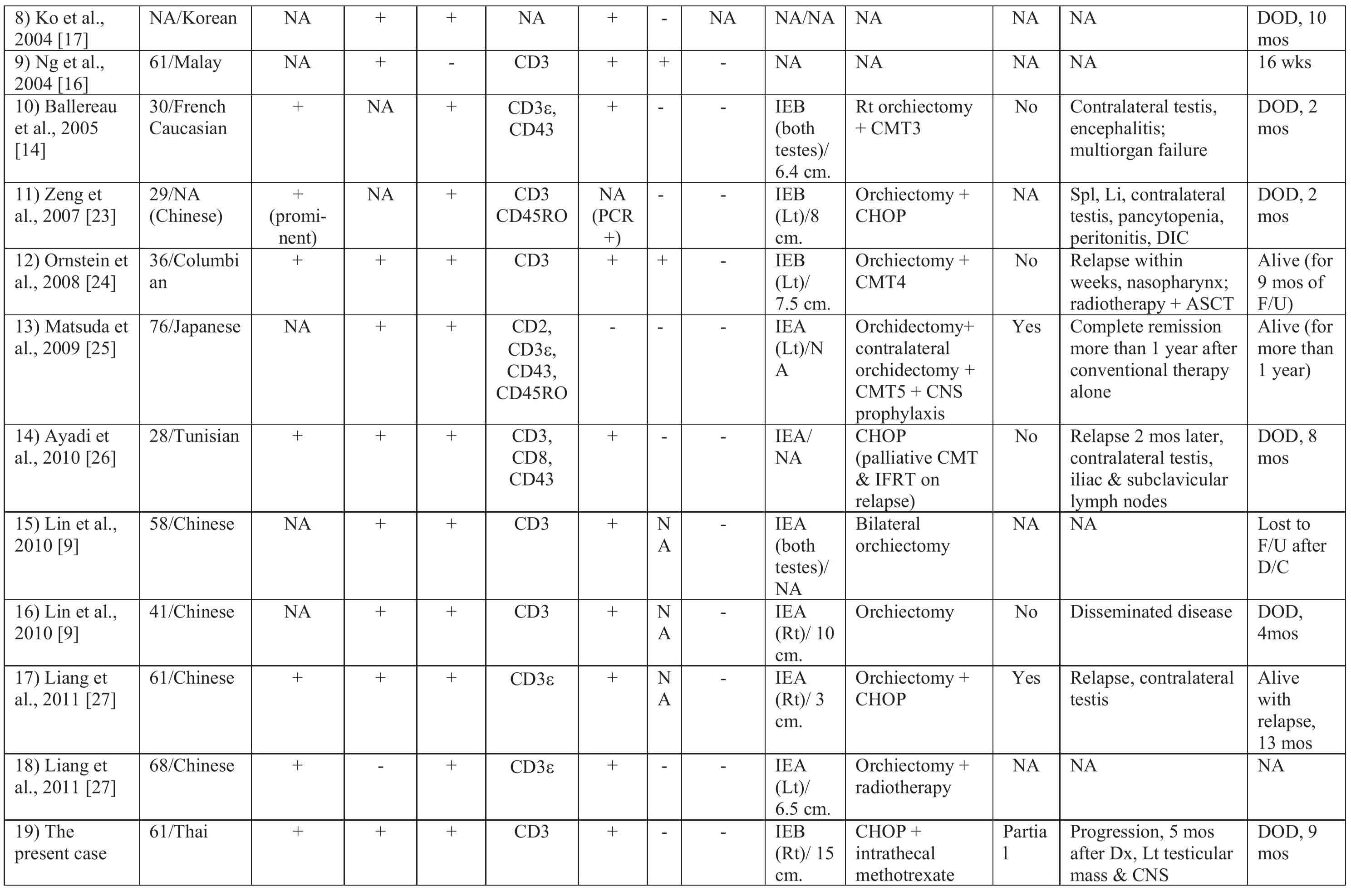
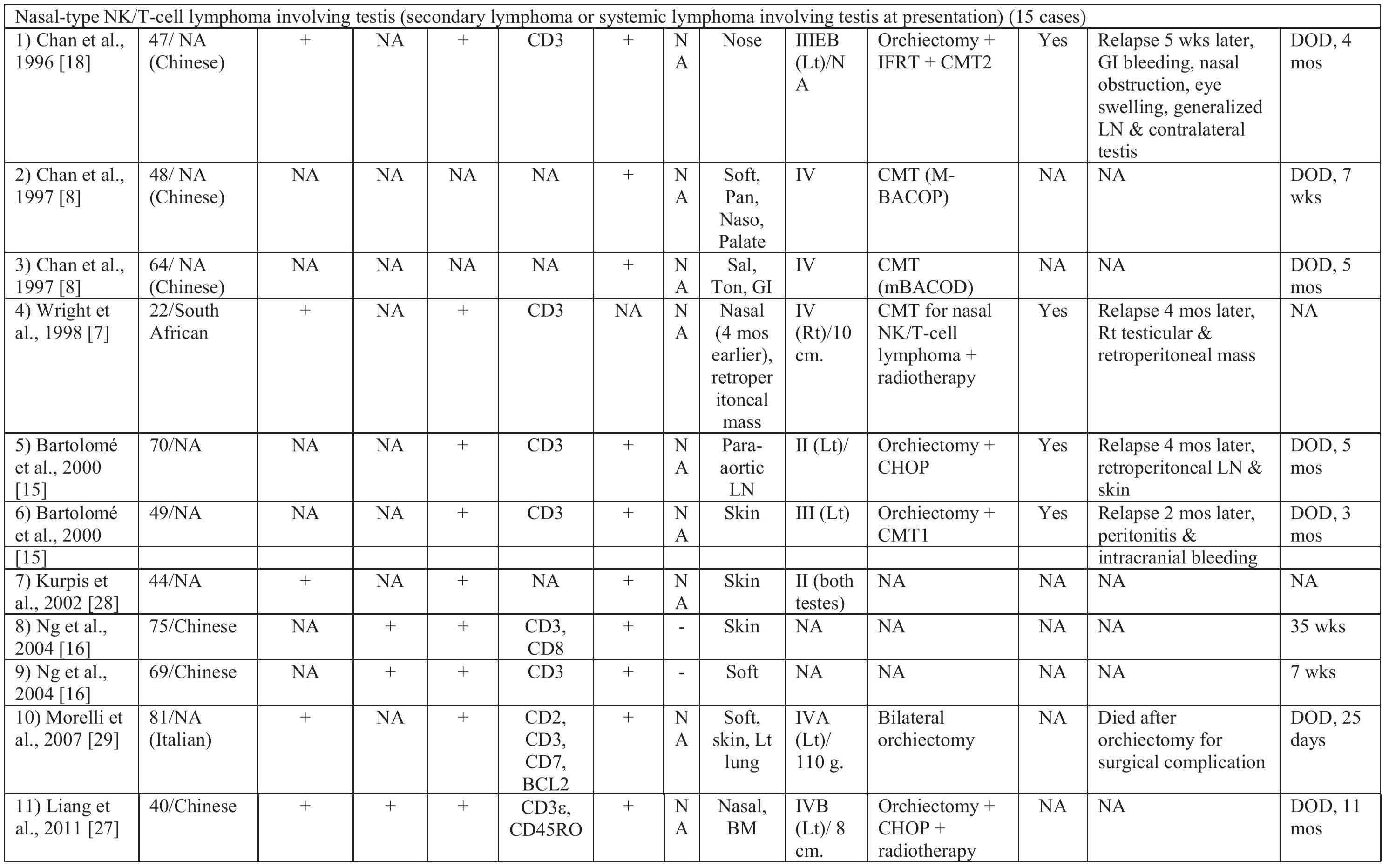
Table 2 Comparison of primary and secondary testicular NK/T-cell lymphoma, nasal type, in the literature
It may be arguable that this is in fact patients with progressive disease, already disseminated at the beginning [18]. A case reported by Sun et al. as aggressive NK-cell lymphoma/leukemia [19] initially presented as primary testicular lymphoma but the disease relapsed 5 months after treatment as leukemic phase. In this particular case, azurophilic granules were described in the lymphoma cells but neither immunostaining for cytotoxic granule associated protein nor in situ hybridization for EBER was performed. Nevertheless, this particular case was often included in the literature review as the first case report of nasal-type NK/T-cell lymphoma of the testis.
In general, the recommended treatment for primary testicular lymphoma of any subtypes includes surgery (orchiectomy) together with local radiotherapy, especially involved field radiotherapy, including the involved scrotum, ipsilateral pelvic area, and/or paraaortic area, and six cycles of an-thracycline-based chemotherapy [3]. Prophylactic radiation to the contralateral testis [11] and prophylactic intrathecal chemotherapy [5] are also recommended. The only case of primary testicular NK/T-cell lymphoma reported by Matsuda et al. [25] was alive more than 1 year at the time of report after a radical orchiectomy plus prophylactic orchiectomy, combination chemotherapy and CNS prophylaxis. Nevertheless, it is difficult to determine whether such an extensive management is appropriate and beneficial to the patient. Among the 5 patients of primary testicular NK/T-cell lymphoma with contralateral testicular relapse [14, 20, 23, 26, 27], all of them received combination chemotherapy but four of them did not receive radiotherapy after orchiectomy while only one patient received radiotherapy. In 15 cases of secondary testicular NK/T-cell lymphoma, only one case received both chemotherapy and radiotherapy but still had contralateral testicular involvement later [18]. It is conceivable that prophylactic radiation to the contralateral testis in patient with primary testicular lymphoma might prevent lymphomatous involvement. In the present case, the progression free survival was 2.5 months (77 days) and the overall survival was 9 months after diagnosis. Certainly, the clinical response to systemic chemotherapy may have been compromised as the tumor burden was not reduced as the patient refused both orchiectomy and local radiotherapy.
Relapse has been reported up to 40% of patients with stage I or II disease of primary nasal NK/T-cell lymphoma who received only local irradiation. Lymphomatous involvement at distant sites such as the skin, gastrointestinal tract and testis is quite common, suggesting occult dissemination in these sites but unable to detect by conventional clinical staging procedure [30]. It is possible that patient who presents with testicular or other primary non-nasal NK/T-cell lymphoma should undergo a thorough ENT examination and even PET/CT scan to search for any occult nasal NK/T-cell lymphoma. If nasal involvement is documented, the patient should receive treatment as advanced disease. The use of L-asparaginase in recent regimens of combination chemotherapy has resulted in substantial improvements in outcome in high-risk, refractory or relapsed patients. High-dose chemotherapy and hematopoietic stem-cell transplantation with autologous or allogeneic hematopoietic stem cells may be beneficial to selected patients. Prognostication of patients with clinical prognostic models, such as modified NK prognostic model, and circulating Epstein-Barr DNA load may be useful in the stratification of patients for various treatment modalities [30].
CONFLICT OF INTEREST
The authors declare that they have no conflict of interest.
ACKNOWLEDGMENTS
All three authors, Drs. Sanya Sukpan-ichnant, Yingyong Chinthammitr, and Naravat Poungvarin, are supported by “Chalermphra-kiat” Grant, Faculty of Medicine Siriraj Hospital, Mahidol University.
REFERENCES
1. Sukpanichnant S. Analysis of 1983 cases of malignant lymphoma in Thailand according to the World Health Organization classification. Hum Pathol 2004;35:224-30.
2. Pongpruttipan T, Sukpanichnant S, Assanasen T, et al. Extranodal NK/T-cell lymphoma, nasal type, includes cases of natural killer cell and ab, y5, and ap/y5 T-cell origin: a comprehensive clinicopathologic and phenotypic study. Am J Surg Pathol 2012;36:481-499.
3. Park B-B, Kim JG, Sohn SK, Kang HJ, Lee SS, Eom HS, et al. Consideration of aggressive therapeutic strategies for primary testicular lymphoma. Am J Hematol 2007;82:840-5.
4. Zucca E, Conconi A, Mughal TI, Sarris AH, Seymour JF, Vitolo U, et al. Patterns of outcome and prognostic factors in primary largecell lymphoma of the testis in a survey by the International Extranodal Lymphoma Study Group. J Clin Oncol 2003;21:20-7.
5. Touroutoglou N, Dimopoulos MA, Younes A, Hess M, Pugh W, Cox J, et al. Testicular lymphoma: Late relapses and poor outcome despite doxorubicin-based therapy. J Clin Oncol 1995;13:1361-7.
6. Swerdlow HS, Campo E, Harris NL, Jaffe ES, Pileri SA, Stien H, et al. World Health Organization Classification of Tumours. Pathology and Genetics of Tumours of Haematopoietic and Lymphoid Tissues. Lyon: IARC Press; 2008.
7. Wright C, Cooper K, Geiman G, Davidge-Pitts M. Natural killer cell lymphoma in cytology: breaking all the rules - a case report. Diagn Cy-topathol 1998;19:9-11.
8. Chan JKC, Sin VC, Wong KF, Ng CS, Tsang WYW, Chan CH, et al. Nonnasal lymphoma expressing the natural killer cell marker CD56: a clinicopathologic study of 49 cases of an uncommon aggressive neoplasm. Blood 1997;89:4501-3.
9. Lin X, Li D, Xie P, Mi C, Lin Q. Primary testicular NK/T-cell lymphoma: a study of two cases and review of literature. Chin J Cancer Res 2010;22:239-44.
10. Ferry JA, Harris NL, Young RH, Coen J, Zi-etman A, Scully RE. Malignant lymphoma of the testis, epididymis, and spermatic cord. A clinicopathologic study of 69 cases with im-munophenotypic analysis. Am J Surg Pathol 1994;18:376-90.
11. Martenson JA Jr, Buskirk SJ, Ilstrup DM, Banks PM, Evans RG, Colgan JP, et al. Patterns of failure in primary testicular non-Hodgkin’s lymphoma. J Clin Oncol 1988;6:297-302.
12. Spears W, Morphis J II, Lester S, Williams SD, Einhorn LH. Brain metastases and testicular tumors: Long-term survival. Int J Radiat Oncol Biol Phys 1991;22:17-22.
13. Kim YB, Chang SK, Yang W-I, Hahn JS, Koom WS, Shim SJ, et al. Primary NK/T cell lymphoma of the testis. A case report and review of the literature. Acta Haematol 2003;109:95-100.
14. Ballereau C, Leroy X, Morschhauser F, Fan-toni J-C, Lemaitre L, Villers A, et al. Testicular natural killer T-cell lymphoma. Int J Urol 2005;12:223-4.
15. Bartolome J, Gonzalez-Arenas M, Menar-guez J, Gil J. Nasal type T/NK cell lymphoma with testicular presentation. [Letter] Med Clin (Barc) 2000;114:318. [Spanish]
16. Ng SB, Lai KW, Murugaya S, Lee KM, Loong SL, Fook-Chong S, et al. Nasal-type extranodal natural killer/T-cell lymphomas: a clinico-pathologic and genotypic study of 42 cases in Singapore. Mod Pathol 2004;17:1097-107.
17. Ko YH, Cho E-Y, Kim J-E, Lee S-S, Huh J-R, Chang H-K, et al. NK and NK-like T-cell lymphoma in extranasal sites: a comparative clin-icopathological study according to site and EBV status. Histopathology 2004;44:480-9.
18. Chan JKC, Tsang WYW, Lau WH, Cheung MMC, Ng WF, Yuen WC, et al. Aggressive T/ natural killer cell lymphoma presenting as testicular tumor. Cancer 1996;77:1198-205.
19. Sun T, Brody J, Susin M, Marino J, Teichberg S, Koduru P, et al. Aggressive natural killer cell lymphoma/leukemia. A recently recognized clinicopathologic entity. Am J Surg Pathol 1993;17:1289-99.
20. Guler G, Altinock G, Uner AH, Sungur A. CD56+ lymphoma presenting as a testicular tumor. Leuk Lymphoma 1999;36:207-11.
21. Perez-Valles A, Sabater-Marco V, Carpio-Manez D, Botella-Estrada R, Nogueira-Vazquez E, Martorell-Cebollada M. Extranodal peripheral T-cell lymphoma with angiocentric growth pattern and Epstein-Barr viral DNA associated affecting paratesticular soft tissue. J Cutan Pathol 2000;27:80-6.
22. Totonchi KF, Engel G, Weisenberg E, Rhone DP, Macon WR. Testicular Natural Killer/T-Cell Lymphoma, Nasal Type, of True Natural Killer-Cell Origin. Arch Pathol Lab Med 2002;126:1527-9.
23. Zeng QB, Cheng FJ, Gao QP, Zhu ZP, Liu QH, Yu Q, et al. Primary NK/T-cell lymphoma of the testis: a case report and review of the literature [J]. Chin-Germ J Clin Oncol 2007;6:596-600.
24. Ornstein DL, Bifulco CB, Braddock DT, Howe JG. Histopathologic and molecular aspects of CD56+ natural killer/T-cell lymphoma of the testis. Int J Surg Pathol 2008;16:291-300.
25. Matsuda M, Iwanaga T, Hashimoto S, Uesugi T, Itagaki N. Primary Epstein-Barr virus-negative nasal-type natural killer.T cell lymphoma of the testis. [Letter to the Editor] Leukemia Research 2009;33:e119-e120. - change to ref.19
26. Ayadi L, Makni S, Toumi N, Hammami S, Charfi S, Frikha M, et al. Aggressive nasal-type natural killer/T-cell lymphoma associated with Epstein Barr virus presenting as testicular tumor. Tunis Med 2010 Mar;88(3):196-8.
27. Liang DN, Yang ZR, Wang WY, et al. Extranodal nasal type natural killer/T-cell lymphoma of testis: report of seven cases with review of literature. Leuk Lymphoma 2012 Jan 3. [Epub ahead of print]
28. Kurpis M, Diller A, Calafat P, Ruiz Lascano A. Extranodal NK/T cell lymphoma of the nasal type. Rev Fac Cien Med Univ Nac Cordoba 2002;59:117-20. [Spanish]
29. Morelli L, Piscioli I, Licci S, Del Nonno F, Neri M, Sabattini E. T/natural killer-cell lymphoma of the testis with cutaneous and subcutaneous soft tissue involvement: a management problem. [Letter to the editor] Ann Hematol 2007;6:469-70.
30. Kwong Y-L. Hematopoietic stem cell transplantation in natural killer cell lymphoma and leukemia. Int J Hematol (2010) 92:702-7.


