Transformation in Malignant Lymphoma:
Morphologic Approach* **
Sanya Sukpanichnant
Department of Pathology, Faculty of Medicine Siriraj Hospital, Mahidol University, Bangkok, Thailand
Telephone: +662-419-6504 (office); +6686-895-0550 (mobile)
Fax: +662-411-4260 E-mail: sanya.suk@mahidol.ac.th
*Partly presented in a lecture on “Histologic Transformation in Lymphoma ” given to the 9th Lymphoma Education Conference
“New Frontiers in Lymphoma” on September 12, 2015 in Bangkok, Thailand,
organized by Thai Lymphoma Study Group (TLSG) under the Thai Society of Hematology.
** Partly presented in text as a review article on “Histologic Transformation in Lymphoma”
published in Thai language in the Journal of Hematology and Transfusion Medicine
on July-September 2015 issue, volume 25, number 3, pages 235-9.
Received 21st september 2015; Accepted 30th October 2015
ABSTRACT
Morphologic approach is still very crucial to detect transformation in malignant lymphoma. With the knowledge of each malignant lymphoma type, especially the clinical course, a number of lymphoma can have histologic progression as evidence of transformation as follow: large cell transformation, blastic transformation, and histiocytic transformation. Pathologists need to be aware of the transformation in lymphoma as the disease develops more aggressive clinical course in most cases and at times it is challenging to distinguish this condition from composite lymphoma and secondary neoplasm. This review together with illustrative examples will emphasize on practical points for morphologic approach upon making diagnosis of transformation in lymphoma.
Keywords: lymphoma transformation • histologic transformation • large cell transformation • Richter transformation • blast transformation • histiocytic transformation
Histologic transformation in malignant lymphoma has been recognized in the literature for decades. It is surprising to learn that the very first case of transformation in lymphoma was published in 19531 - seven years prior to our understanding about lymphocyte transformation that was published in 1960 when phytohemagglutinin was used as an initiator of mitosis in cultures of normal human leukocytes2.
Among the common types of lymphoma with transformation at first presentation or later during several years of follow-up are follicular lymphoma (FL), small lymphocytic lymphoma (SLL)/chronic lymphocytic leukemia (CLL), and mycosis fungoides (MF). All these 3 lymphomas are regarded as indolent or low grade lymphoma. So, when transformation occurs in these lymphomas, the patients mostly show more aggressive clinical courses. This phenomenon, thus, raises the attention among the clinicians who take care of the patients. Certainly, it has been addressed in these 3 lymphomas and other types in the 2008 version of WHO classification for lymphoid neoplasms1 2 3.
Transformation in lymphoma has been regarded as morphologic or histologic progression of the lymphoma cells as the transformed lymphoma cells are presumably derived from the original lymphoma cells. Later, advances in immunopheno-typic and genetic studies provide sufficient evidence that, at the time of transformation, the transformed lymphoma cells are different from the original lymphoma cells. Thus, the term “transformation” has been discarded in the WHO classification (2008) - for example, instead of large cell transformation in follicular lymphoma, the diagnosis is given as diffuse large B-cell lymphoma with accompanying follicular lymphoma and the corresponding areas of involvement by each type of lymphoma are provided3.
Nevertheless, morphologic approach is still crucial when malignant lymphoma is diagnosed before further investigations are considered for immunophenotypic and genetic studies. This review will emphasize the importance of morphologic approach by showing illustrative examples to pathologists upon the practical points regarding transformation in lymphoma.
There are 3 main types of transformation in lymphoma, including large cell, blastic, and histiocytic transformation.
lymphoma cells are typical Hodgkin-Reed-Stern-berg (HRS) cells and their variants. These HRS cells commonly show immunophenotypic features described in classical Hodgkin lymphoma (CHL). In practice, it is important to distinguish between CHL and Hodgkin transformation in SLL/CLL or other types of lymphoma. Lymphocyte-rich CHL at times creates problem as its common nodular variant contains predominantly small B-cells of mantle cell phenotype (IgM+ IgD+) but, in daily practice, evaluation of both IgM and IgD expression is hardly determined, especially in countries with limited resources. Immunophenotype of lymphoma cells in SLL/CLL may be difficult to distinguish from small B-cells of mantle cell phenotype described in lymphocyte-rich CHL. In this situation, nodules are important to distinguish between lymphocyte-rich CHL and SLL/CLL with Hodgkin transformation because they are lacking in SLL/CLL. But difficulty may emerge when core needle biopsy is performed, thus precluding the appreciation of the nodules. Certainly, previous history of SLL/CLL should ease such a distinction. Another practical point is the recognition of proliferation centers (the so-called “pseudofollicles”) in typical SLL/CLL and one should keep in mind that proliferation centers are much smaller than nodules in lymphocyte-rich CHL3. (Figure 1)
Large cell transformation is more common to be non-Hodgkin type even though HRS cells may be occasionally observed. The transformed lymphoma cells are definitely large with their nuclear sizes frequently larger than those of histiocytes. They may be centroblasts, immunoblasts, plasmablasts, anaplastic large cells or hallmark cells described in anaplastic large cell lymphoma. The criterion provided in the WHO classification (2008) is the presence of sheet of large B-cells for making diagnosis of diffuse large B-cell lymphoma (DLBCL) when occurs in the background of indolent lymphomas. Unfortunately, the size of this sheet has never been defined.
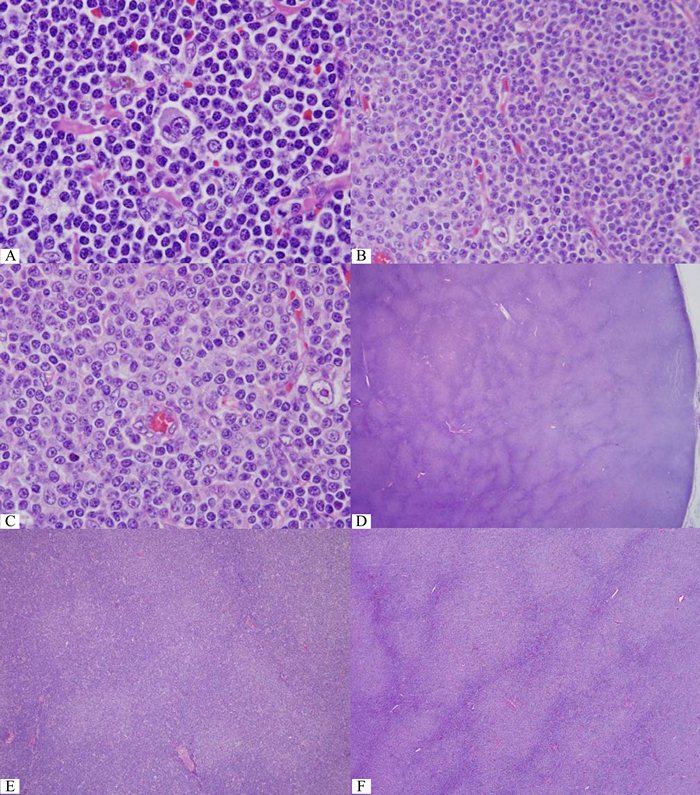
Figure 1 One Reed-Sternberg like cell in small lymphocytic lymphoma/chronic lymphocytic leukemia (SLL/CLL). B&C) One Hodgkin cell abut typical proliferation center in SLL/CLL. D) Pseudofollicles in SLL/CLL. Note the coalescent areas that require careful evaluation for large cell transformation. E) Separate proliferation centers typically seen in SLL/CLL. F) Coalesced proliferation centers should raise the concern of large cell transformation. G to I) An unusually large proliferation center. At higher magnification, only larger cells are increased (H) and some para-immunoblasts (equivalent to prolymphocytes described by hematologists) (I) but not solid sheet enough for large cell transformation. J&K) Typical Ki-67+ cells within each proliferation center and only a small number of Ki-67+ cells outside these centers. (Compare to E) L&M) More Ki-67+ cells, especially some large ones, in the coalesced proliferation centers but no solid sheet of Ki-67+ cells for large cell transformation. (Compare to F to I)
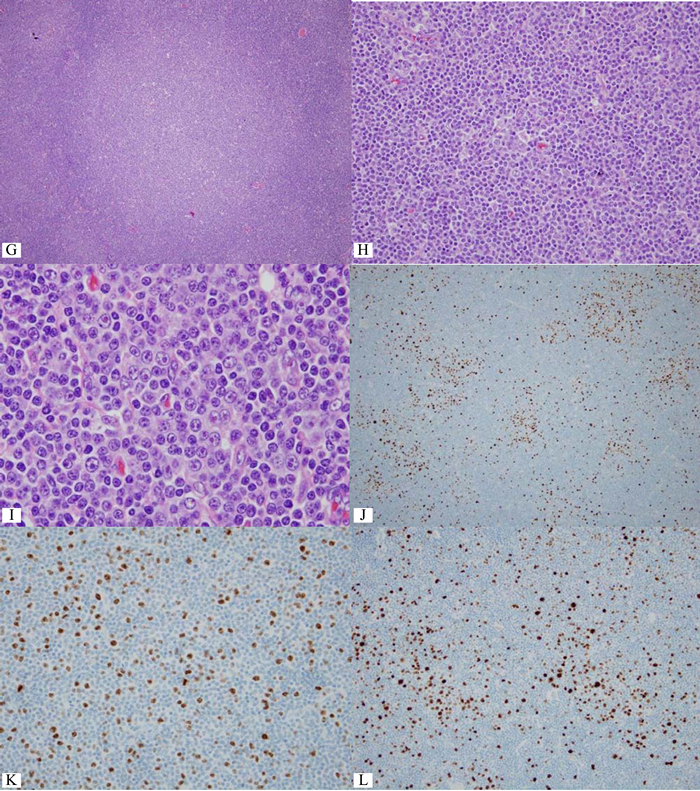
Figure 1 One Reed-Sternberg like cell in small lymphocytic lymphoma/chronic lymphocytic leukemia (SLL/CLL). B&C) One Hodgkin cell abut typical proliferation center in SLL/CLL. D) Pseudofollicles in SLL/CLL. Note the coalescent areas that require careful evaluation for large cell transformation. E) Separate proliferation centers typically seen in SLL/CLL. F) Coalesced proliferation centers should raise the concern of large cell transformation. G to I) An unusually large proliferation center. At higher magnification, only larger cells are increased (H) and some para-immunoblasts (equivalent to prolymphocytes described by hematologists) (I) but not solid sheet enough for large cell transformation. J&K) Typical Ki-67+ cells within each proliferation center and only a small number of Ki-67+ cells outside these centers. (Compare to E) L&M) More Ki-67+ cells, especially some large ones, in the coalesced proliferation centers but no solid sheet of Ki-67+ cells for large cell transformation. (Compare to F to I)
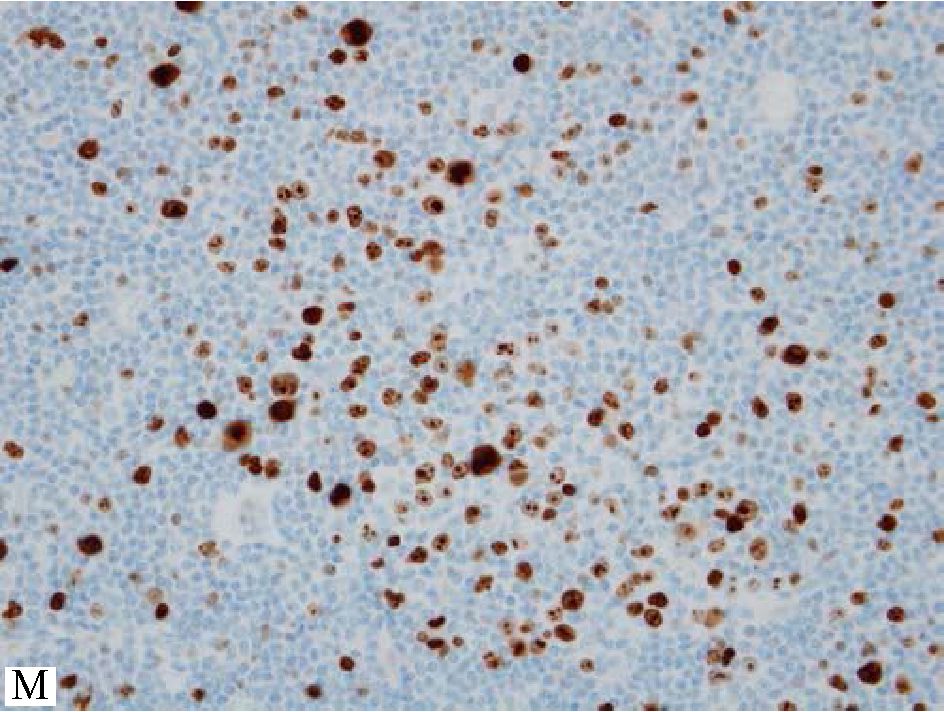
Figure 1 One Reed-Sternberg like cell in small lymphocytic lymphoma/chronic lymphocytic leukemia (SLL/CLL). B&C) One Hodgkin cell abut typical proliferation center in SLL/CLL. D) Pseudofollicles in SLL/CLL. Note the coalescent areas that require careful evaluation for large cell transformation. E) Separate proliferation centers typically seen in SLL/CLL. F) Coalesced proliferation centers should raise the concern of large cell transformation. G to I) An unusually large proliferation center. At higher magnification, only larger cells are increased (H) and some para-immunoblasts (equivalent to prolymphocytes described by hematologists) (I) but not solid sheet enough for large cell transformation. J&K) Typical Ki-67+ cells within each proliferation center and only a small number of Ki-67+ cells outside these centers. (Compare to E) L&M) More Ki-67+ cells, especially some large ones, in the coalesced proliferation centers but no solid sheet of Ki-67+ cells for large cell transformation. (Compare to F to I)
In SLL/CLL, Richter transformation is commonly used when large cell transformation occurs and the clinical course often gets worse; the survival after transformation to large cell lymphoma is less than a year in the literature.3 Richter transformation occurs in 2-15% of patients with CLL and, in approximately half of them, it occurs within the period of 2-4 years. Most of Richter transformation cases (90%) are DLBCL while the rest are Hodgkin type as already mentioned4-6. Median survival in the former is 8-12 months while the latter 20 months5. Regarding the cell of origin, activated B-cell phenotype is often found among the transformed large cells and many cases have been successfully shown that they probably originate from the neoplastic cells in CLL.
Evidence from molecular genetic studies demonstrates abnormal regulation in cell cycle, especially the inactivation of TP53 or CDKN2A genes, in approximately half of the patients with Richter transformation of DLBCL type while one-third of the patients related to trisomy 124.
The problem in practice is how to distinguish large cell transformation from proliferation center that is described as “a continuum of small, medium, and large cells. Prolymphocytes are small to medium-sized cells with relatively clumped chromatin and small nucleoli; paraimmunoblasts are larger cells with round to oval nuclei, dispersed chromatin, central eosinophilic nucleoli and slightly basophilic cytoplasm. The size of proliferation centers and number of paraimmunoblasts vary from case to case, but there is no correlation between lymph node histology and clinical course. Over time, CLL may show an increase in cell size and proliferative activity as well as confluence of proliferation centers in lymph nodes and BM (bone marrow). Often, this may correlate with an increase in prolymphocytes in the PB (peripheral blood). Progression of CLL to B-PLL (B-cell prolymphocytic leukemia) is extremely rare. 2-8% of patients with CLL develop DLBCL and <1% develop CHL.3” The problem here is how large the “confluence of proliferation centers” is when examined under microscope. (Figure 1)
The only statement concerning the diagnosis of DLBCL in the background of indolent lymphoma in the WHO classification (2008) is in the chapter of FL as “diffuse areas containing >15 centroblasts per hpf are reported as DLBCL with follicular lymphoma (Grade 1-2, Grade 3A, or Grade 3B) and give approximate % of each in report.3” The practical point is to evaluate only the diffuse areas, not the neoplastic follicles, and count the number of centroblasts to decide whether there is any evidence of DLBCL or not. (Figure 2) Again, difficulty arises when the sample is obtained by core needle biopsy that precludes the precise distinction between neoplastic follicles and diffuse areas.
Another important practical point is about careful interpretation of Ki-67 when applied to evaluate proliferation index in FL. At times, residual reactive germinal centers may be found within the lymphoma areas and it should not lead to overgrading FL when evaluating Ki-67 solely without awareness of this fact. Comparison with BCL2 protein expression of the high Ki-67+ focus on the immu-nostained section for BCL2 protein just next to the immunostained section for Ki-67 is very important as BCL2 protein expression should be lacking if this high Ki-67+ focus is in fact residual reactive germinal center. (Figure 3)
Clinically, FL has been one of the oldest lymphoma known to have transformation. In the past, without immunophenotypic evidence, approximately 30-40% of the patients with FL developed large cell transformation and half of them developed 5 years after the diagnosis of FL was given. No clinical or morphologic features were found as a predictor for large cell transformation. Whenever the patients develop clinical discomforts or enlarging lymph node despite specific treatment, biopsy of the lymph node should be performed to obtain tissue for evidence of large cell transformation78. A recent interesting study about the natural clinical course of FL in patients who chose to receive no treatment, just only “watch and wait,” the cumulative risk of histologic transformation within 10 years was 22% and the 3 year overall survival rate after transformation was 71%9. While another study using rituximab-based regimen, 4.5% of the patients with FL had large cell transformation within 5 years and 5 year overall survival rate after transformation was 50%10.
Advance in technology provides new insight for our understanding of large cell transformation in FL such as altered immune surveillance, nuclear factor-kappaB pathway stimulation, decreased control of p53 and factors involving B-cell transcription, etc. Recently, using gene expression profiling (GEP) technique, transformed lymphoma cells in FL have been proven about their germinal center B-cell phenotype as expected. But, interestingly, approximately 20% of them showed activated B-cell phenotype by GEP11. Another study also supports this observation and proposed expression of IRF4/ MUM1 as a predictor for early transformation and mixed morphologic features of both DLBCL and FL at the time of transformation as favorable histology. The association between the lack of BCL2 translocation of FL at diagnosis and the transformation of activated B-cell like large cell lymphoma was observed12.
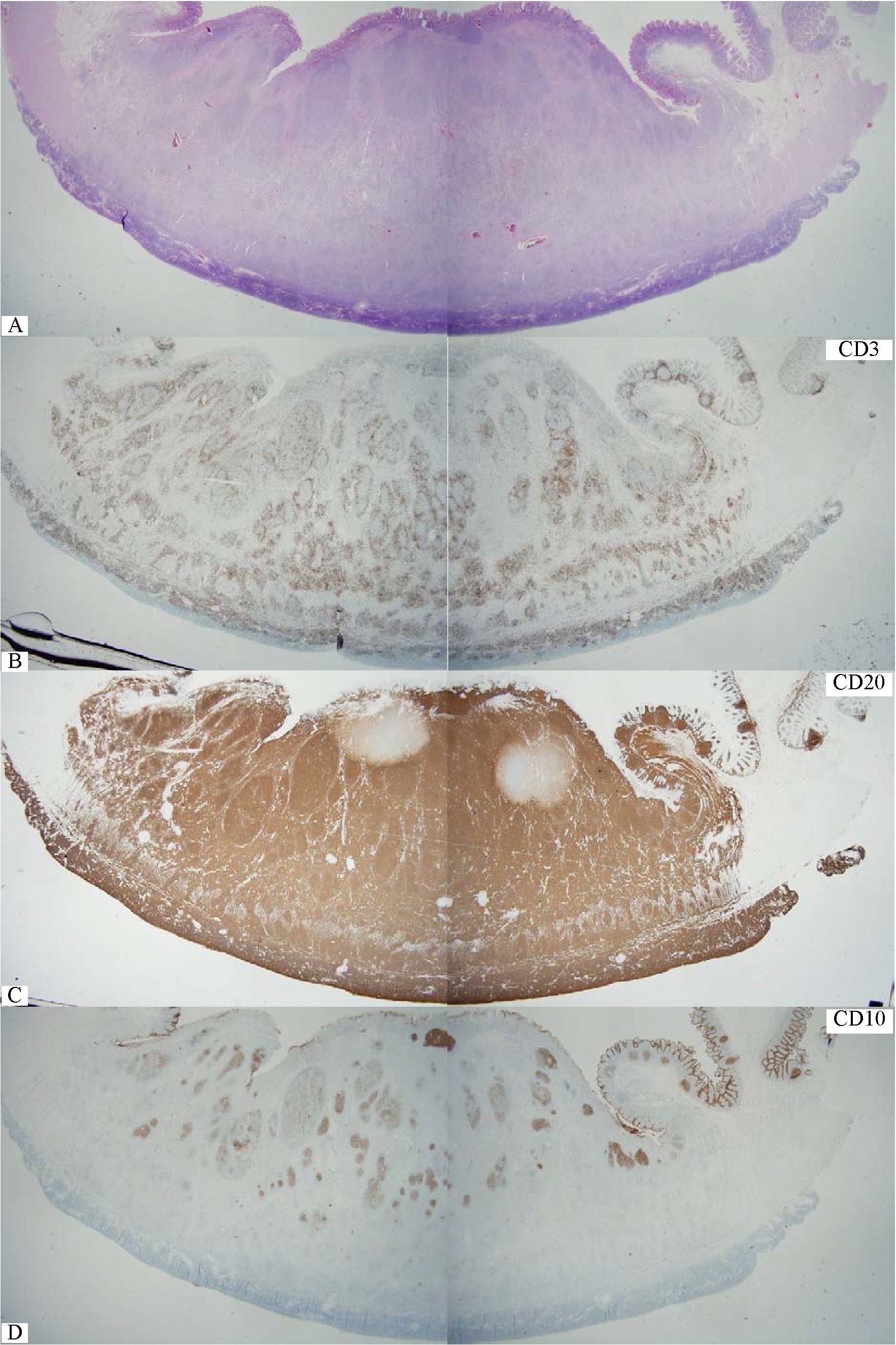
Figure 2 Whole section view of follicular lymphoma of the small intestine. Note definite neoplastic follicles even at this low magnification. B) CD3+ reactive small T-cells outline each neoplastic follicle. C) CD20+ follicular lymphoma cells infiltrates throughout the thickness of the bowel wall. D) CD10+ lymphoma cells are confined in some neoplastic follicles. E) BCL2+ follicular lymphoma. Note in situ follicular lymphoma in the mucosa nearby (red box). F) Compilation of various markers for in situ follicular lymphoma. G&H) Diffuse area with many large lymphoma cells, sufficient for large cell transformation.
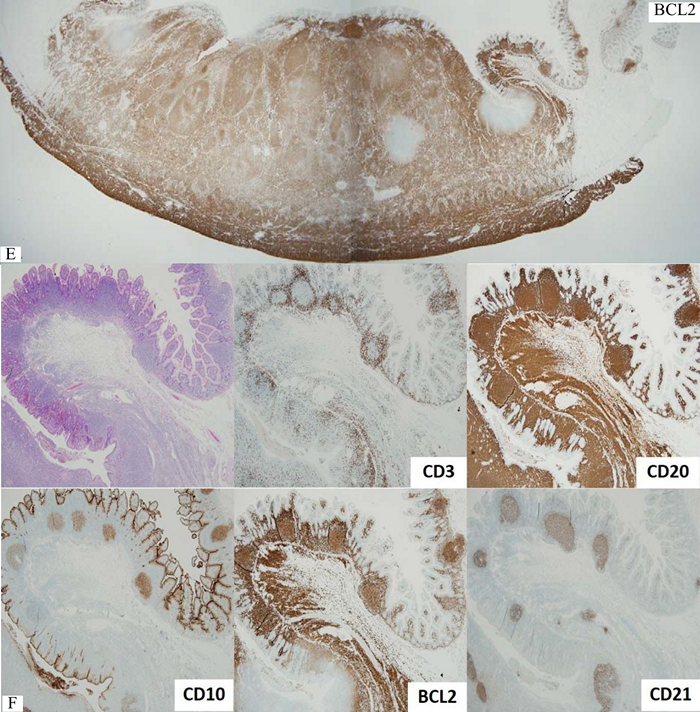
Figure 2 Whole section view of follicular lymphoma of the small intestine. Note definite neoplastic follicles even at this low magnification. B) CD3+ reactive small T-cells outline each neoplastic follicle. C) CD20+ follicular lymphoma cells infiltrates throughout the thickness of the bowel wall. D) CD10+ lymphoma cells are confined in some neoplastic follicles. E) BCL2+ follicular lymphoma. Note in situ follicular lymphoma in the mucosa nearby (red box). F) Compilation of various markers for in situ follicular lymphoma. G&H) Diffuse area with many large lymphoma cells, sufficient for large cell transformation.
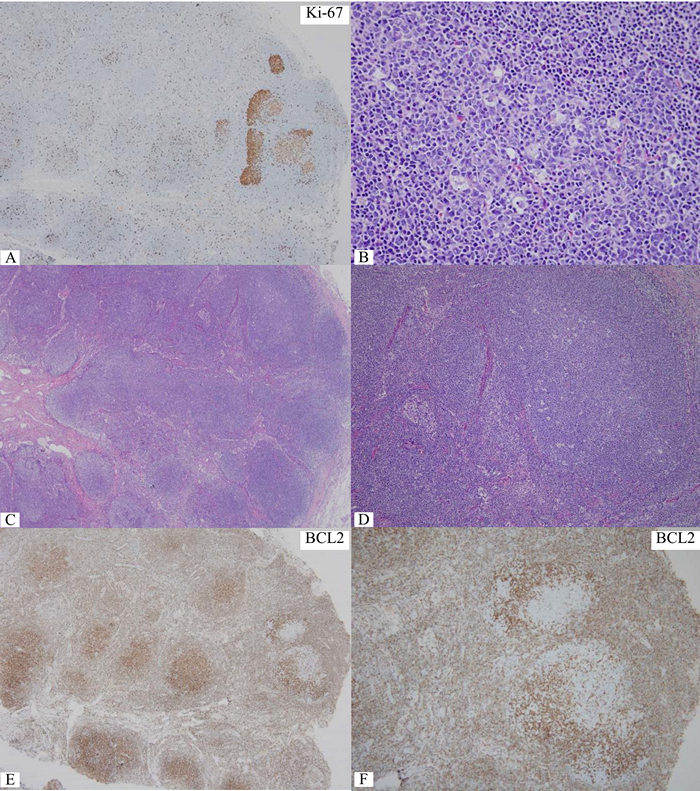
Figure 3 Ki-67+ cells accumulate in small foci at right upper corner while only a small number in other areas, query foci of grade 3 or large cell transformation. B) Significantly increase in centroblasts in the corresponding focus. C) Low magnification of the same node, the focus of interest is still at right upper corner. D) Medium magnification of the focus of interest. With lower magnification in C&D, one can appreciate polarity or zoning phenomenon in reactive germinal centers. E&F) Lack of BCL2 protein in the focus of interest at corresponding magnification to C&D, confirming reactive germinal centers. One should be aware of this pitfall to avoid overgrading or even large cell transformation.
Another common lymphoma in daily pathology practice is gastric lymphoma, especially extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue (MALT lymphoma). The WHO classification mentions only one short statement at the end of the chapter of MALT lymphoma as “transformation to DLBCL may occur.” But, when concerning about morphology, the WHO classification describes MALT lymphoma as follow: “MALT lymphoma as defined is a lymphoma composed predominantly of small cells. Transformed centro-blast- or immunoblast-like cells may be present in variable numbers in MALT lymphoma but, when solid or sheet-like proliferations of transformed cells are present, the tumor should be diagnosed as DLBCL and the presence of accompanying MALT lymphoma noted. The term “high-grade MALT lymphoma” should not be used, and the term “MALT lymphoma” should not be applied to a large B-cell lymphoma even if it has arisen in a MALT site or is associated with lymphoepithelial lesions.3” In daily pathology practice, the endoscopic biopsy of gastric mucosa is obviously different from gastrectomy specimen. When transformed lymphoid cells are present in the abnormal dense lymphoid infiltration that is suspicious of malignant lymphoma, it is crucial to determine whether this is MALT lymphoma or DLBCL. Certainly, MALT lymphoma with increased number of large B-cells is always problematic as the WHO classification (2008) never defines how large “the solid or sheet-like proliferations of transformed cells” is. It is not certain whether we can use the same criterion just mentioned previously in DLBCL with follicular lymphoma as >15 centroblasts per hpf in the background of MALT lymphoma for making diagnosis of DLBCL with MALT lymphoma. The other option is about using the criterion of the so-called “high-grade MALT lymphoma” as at least 20 large B-cells per hpf13. Immunostaining for Ki-67+ transformed cells is very helpful for better detection of Ki-67+ cells whether they form aggregate or not. The proliferation fraction or proliferation index (PI) in typical MALT lymphoma is expected to be low but no definite number is given. But, one may use the PI in low grade of follicular lymphoma that is less than 20% of the lymphoma cells3.
Large cell transformation in mycosis fungoides (MF) is frequently an issue of concern. It can occur in 20-50% of patients with MF and median survival is not more than 12 months. Extracutaneous sites of large cell transformation, such as lymph nodes, can occur and the clinical course may be worse. International Prognostic Index (IPI) is still feasible while other histologic features have been evaluated as prognostic implication such as presence of follicular mucin, lack of papillary dermal involvement, and lack of dermal fibrosis14-16.
It should not be a problem when large cell transformation occurs in the known case of MF even though the criterion about the number of large cells per hpf or the exact size of sheet of large cells has never been defined3. The difficult situation is when large cell transformation occurs simultaneously with MF at first presentation. The most important differential diagnosis is anaplastic large cell lymphoma (ALCL) that involves the epidermis. CD30 expression can be observed in transformed large cells in MF, usually admixed with smaller lymphoma cells. But, when the CD30 expression is found in at least 75% of the large cells, it is not possible to distinguish large cell transformation in MF from ALCL based on the CD30 expression alone17. A large panel of immunostaining may be helpful to distinguish between these two entities despite the fact that both have CD4 expression.
Cytotoxic granule associated proteins, such as TIA-1 and granzyme B, are expressed in ALCL but they should be negative in large cell transformation in MF. Regarding ALK protein expression, it is useful to distinguish between primary cutaneous ALCL and cutaneous involvement by systemic ALK+ ALCL. Recently, a rare case of ALK+ CD30+ MF (reported under the name of primary cutaneous T-cell lymphoma) has been reported18. So, it is important to have a complete clinical information and clinical work-up for a definite diagnosis when encountered with large cell transformation of MF that shows CD30 expression. (Figure 4)
Other types of large cell transformation are uncommon but may cause problem for making a definite diagnosis. For example, in plasmablastic transformation, the transformed lymphoma cells resemble plasmablasts or immunoblasts and at times it is not possible to use the term correctly. As a matter of fact, immunoblastic sarcoma was used in the past as a morphologic variant of the so-called “reticulum cell sarcoma” and the term “immunoblast” was used to make it different from centroblast. While the term “plasmablast” was first used in multiple myeloma and it has been regarded as one of the poor prognostic factors. During that time, immunoblastic sarcoma was needed to distinguish from plasmacytoma or multiple myeloma with increased plasmablasts as they are different clinically. But, at present, according to the WHO classification (2008), the equivalent type of lymphoma for immunoblastic sarcoma seems to be plasmablastic lymphoma while the term “immunoblastic” is only found as a morphologic variant in DLBCL that still have typical immunophe-notype, especially CD20 expression3. At times, it is still difficult to distinguish between plasmablastic lymphoma and plasmacytoma/plasma cell myeloma with increased plasmablasts - similar to what happened to immunoblastic sarcoma in the past. In this very situation, clinical information is very helpful to reach a definite diagnosis. (Figure 5)
The term “plasmacytic differentiation” has not been included in histologic transformation in lymphoma despite the fact that it is clearly a transformation, not only morphologically but also im-munophenotypically. It was included at least once, nevertheless, in a thorough review by York et al. in 19847. The reason behind this plasmacytic differentiation not included from the transformation in lymphoma is that not only lymphoplasmacytic lymphoma but also other indolent B-cell lymphomas can have plasmacytic differentiation. Even in DLBCL, some cases may show restriction to light chain or heavy chain of immunoglobulin as a result of plasmacytic differentiation. But this phenomenon does not make the disease more aggressive but only some consequences related to excess immunoglobulin production such as autoimmune hemolytic anemia, polyneuropathy, light chain deposition disease, amyloidosis, etc. In daily pathology practice, this proven plasmacytic differentiation can be used to monitor the disease such as minimal involvement and to distinguish it from reactive response, especially after treatment. In rare occasion, plasmacytic differentiation may be the only finding found in the tissue biopsy and it is very difficult to distinguish it from plasmacytic neoplasm or plasma cell myeloma. Clinical correlation is a must in this situation to solve the problem.
Since the understanding of Hodgkin-Reed-Sternberg (HRS) cells in CHL in the past decade has increased tremendously due to advance in molecular genetics, the origin of HRS cells is now accepted to be germinal center B-cell phenotype in most cases of CHL19. The recently recognized entity of unclassifiable B-cell lymphoma with intermediate features between CHL and DLBCL listed in the WHO classification (2008)3 is another intriguing issue related to large cell transformation when expression of both CD20 and CD30 is detected while the other immunophenotypic findings may be admixed between CHL and DLBCL phenotypes (Hodgkin-like large B-cell transformation). (Figure 6)
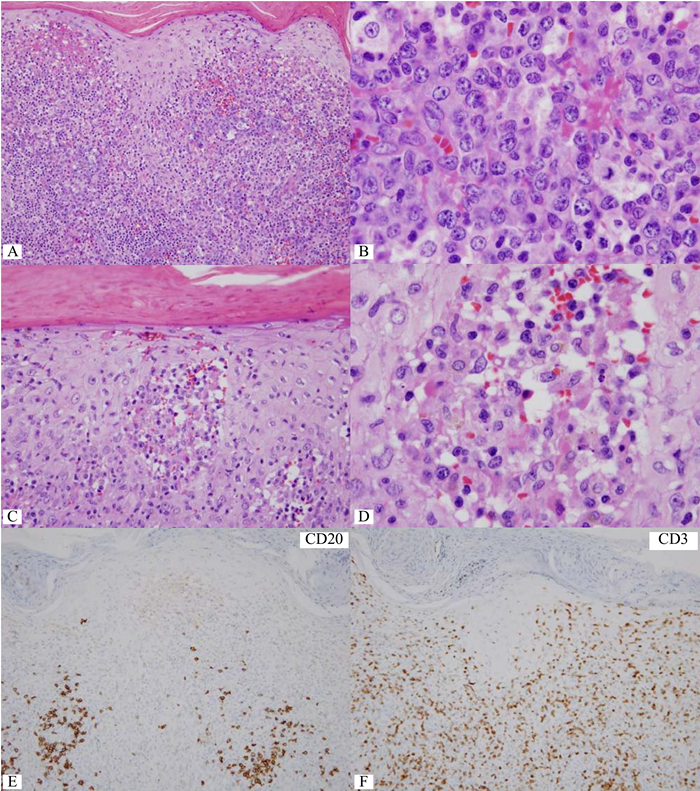
Figure 4 Destruction of dermo-epidermal junction by lymphoma cells with increase in large lymphoma cells.
B) Dense focus of large lymphoma cells. C&D) Collection of lymphoma cells of varying sizes in the papillary dermis, mimicking Pautrier micro-abscess. By morphology alone, large cell transformation in mycosis fungoides (MF) is considered. E) CD20-negative lymphoma cells. F) A number of CD3+ lymphoid cells but lacking in large cells. G&H) Many CD30+ large lymphoma cells. I) Nuclear and cytoplasmic expression of ALK protein in the lymphoma cells. This case should be diagnosed as cutaneous involvement by ALK+ anaplastic large cell lymphoma, thus excluding large cell transformation in MF.
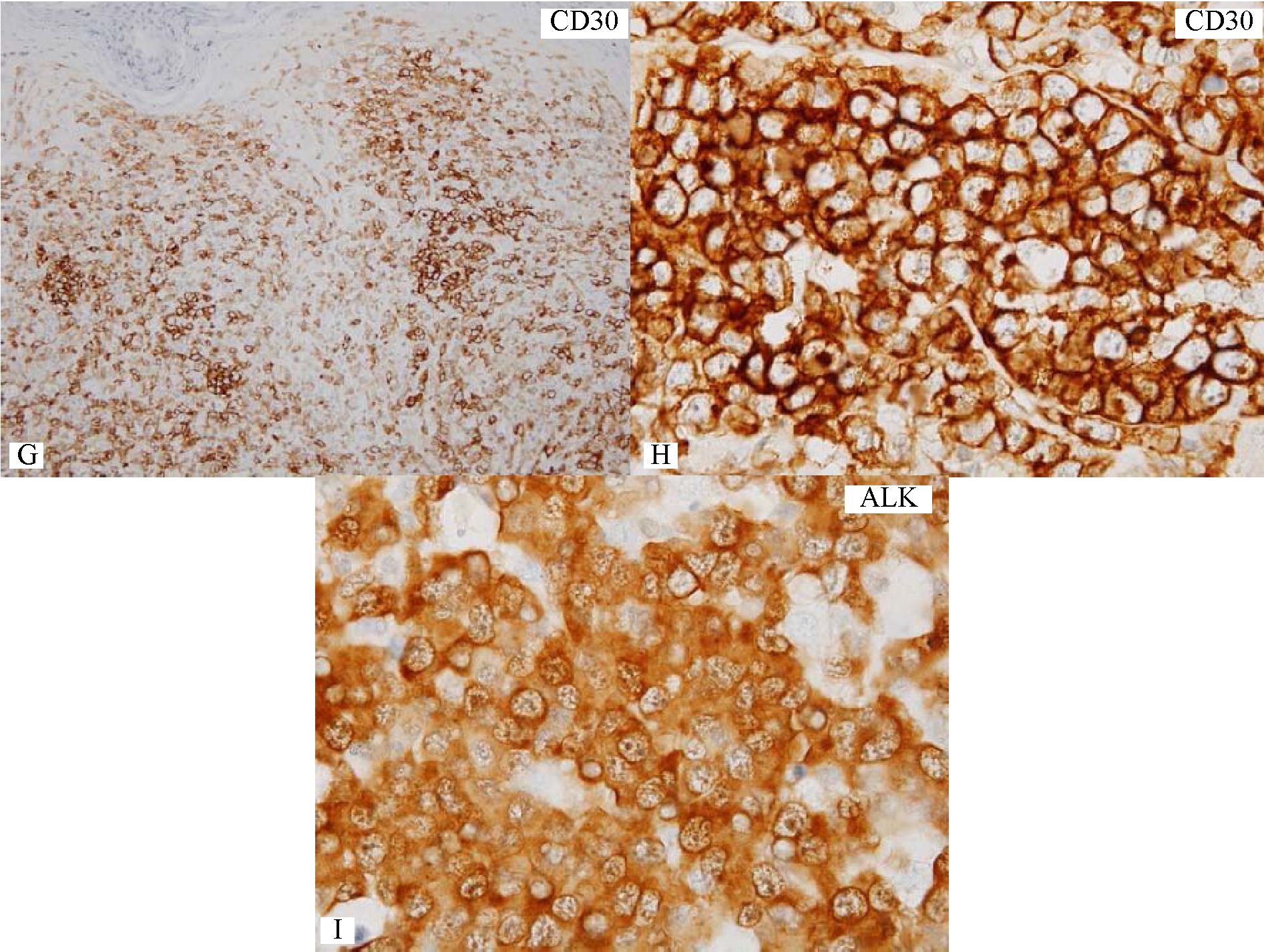
Figure 4 Destruction of dermo-epidermal junction by lymphoma cells with increase in large lymphoma cells.
B) Dense focus of large lymphoma cells. C&D) Collection of lymphoma cells of varying sizes in the papillary dermis, mimicking Pautrier micro-abscess. By morphology alone, large cell transformation in mycosis fungoides (MF) is considered. E) CD20-negative lymphoma cells. F) A number of CD3+ lymphoid cells but lacking in large cells. G&H) Many CD30+ large lymphoma cells. I) Nuclear and cytoplasmic expression of ALK protein in the lymphoma cells. This case should be diagnosed as cutaneous involvement by ALK+ anaplastic large cell lymphoma, thus excluding large cell transformation in MF.
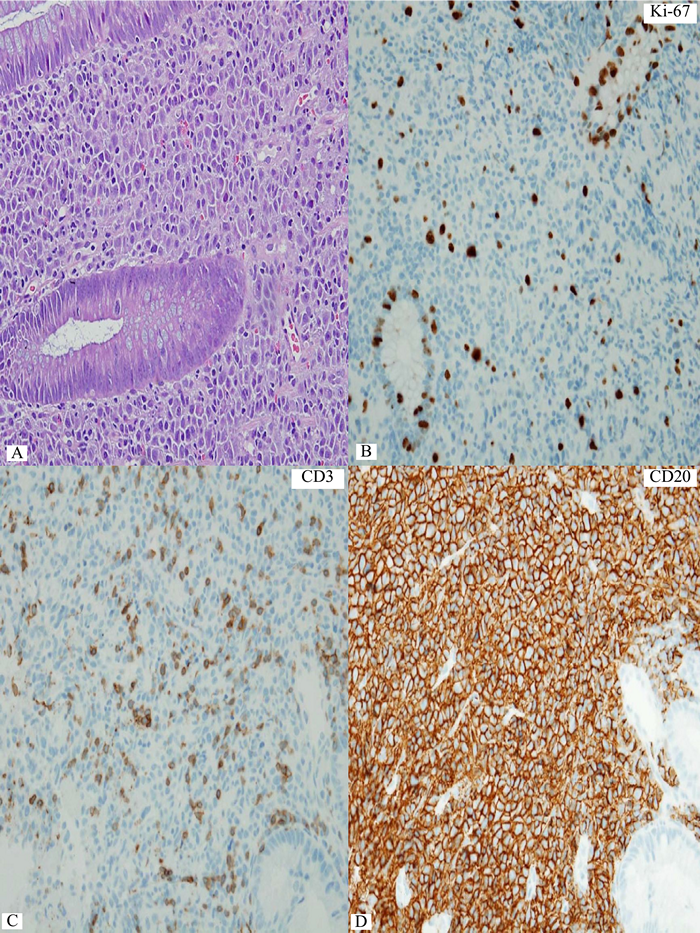
Figure 5 Rectal lymphoma showing some varying sizes of lymphoma cells. B) Only a small number of scattered Ki-67+ lymphoma cells guarantee low proliferation index in this lymphoma. C) A small number of CD3+ small reactive T-cells. D to F) Distinct expression of CD20, PAX5, and CD138 in the lymphoma cells. G&H) Restriction to cytoplasmic kappa light chain indicates plasmcytic differentiation in the lymphoma. I&J) Increase in large lymphoma cells in small clusters and aggregates on follow-up biopsy. K&L) Coexpression of CD20 and CD138 still persists at the time of large cell (plasmablastic) transformation
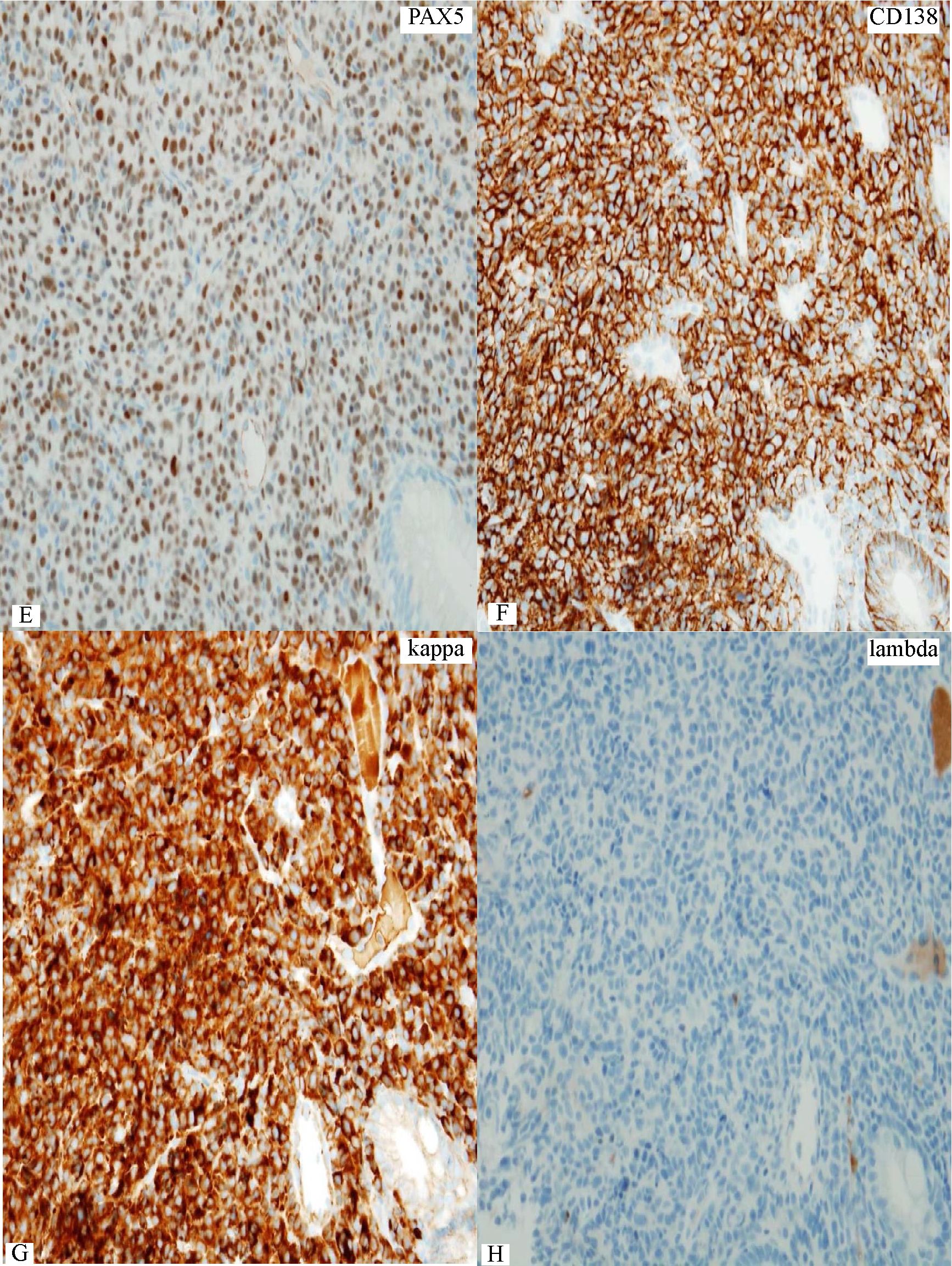
Figure 5 Rectal lymphoma showing some varying sizes of lymphoma cells. B) Only a small number of scattered Ki-67+ lymphoma cells guarantee low proliferation index in this lymphoma. C) A small number of CD3+ small reactive T-cells. D to F) Distinct expression of CD20, PAX5, and CD138 in the lymphoma cells. G&H) Restriction to cytoplasmic kappa light chain indicates plasmcytic differentiation in the lymphoma. I&J) Increase in large lymphoma cells in small clusters and aggregates on follow-up biopsy. K&L) Coexpression of CD20 and CD138 still persists at the time of large cell (plasmablastic) transformation
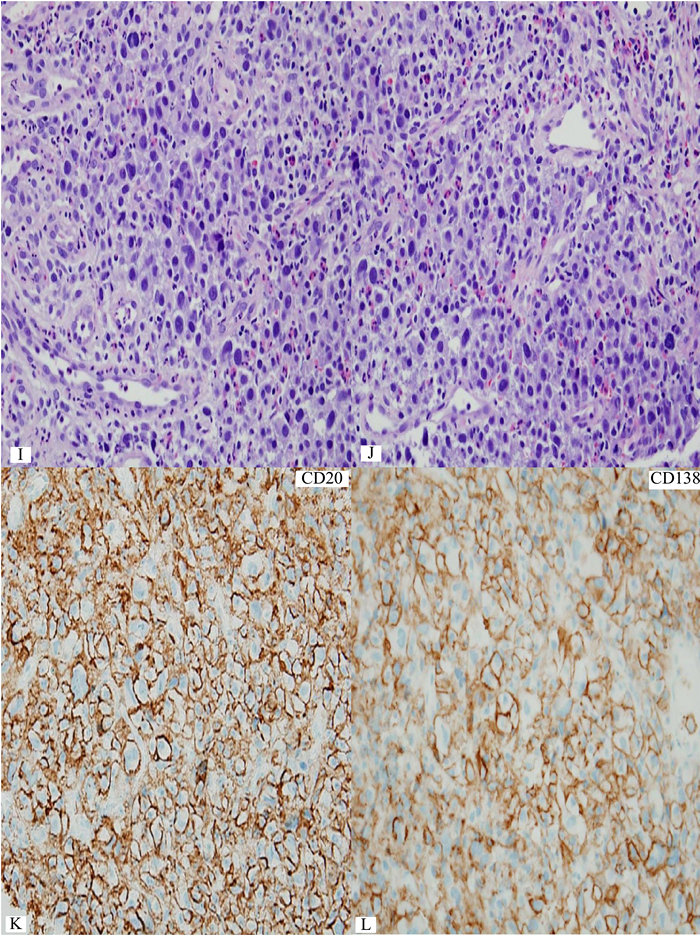
Figure 5 Rectal lymphoma showing some varying sizes of lymphoma cells. B) Only a small number of scattered Ki-67+ lymphoma cells guarantee low proliferation index in this lymphoma. C) A small number of CD3+ small reactive T-cells. D to F) Distinct expression of CD20, PAX5, and CD138 in the lymphoma cells. G&H) Restriction to cytoplasmic kappa light chain indicates plasmcytic differentiation in the lymphoma. I&J) Increase in large lymphoma cells in small clusters and aggregates on follow-up biopsy. K&L) Coexpression of CD20 and CD138 still persists at the time of large cell (plasmablastic) transformation
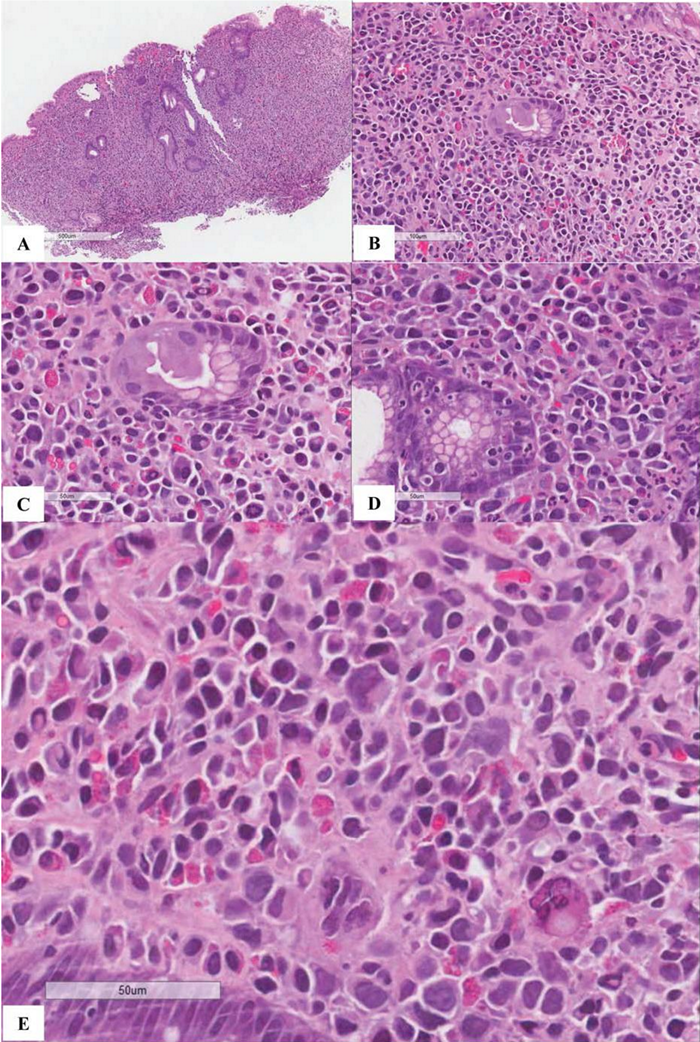
Figure 6
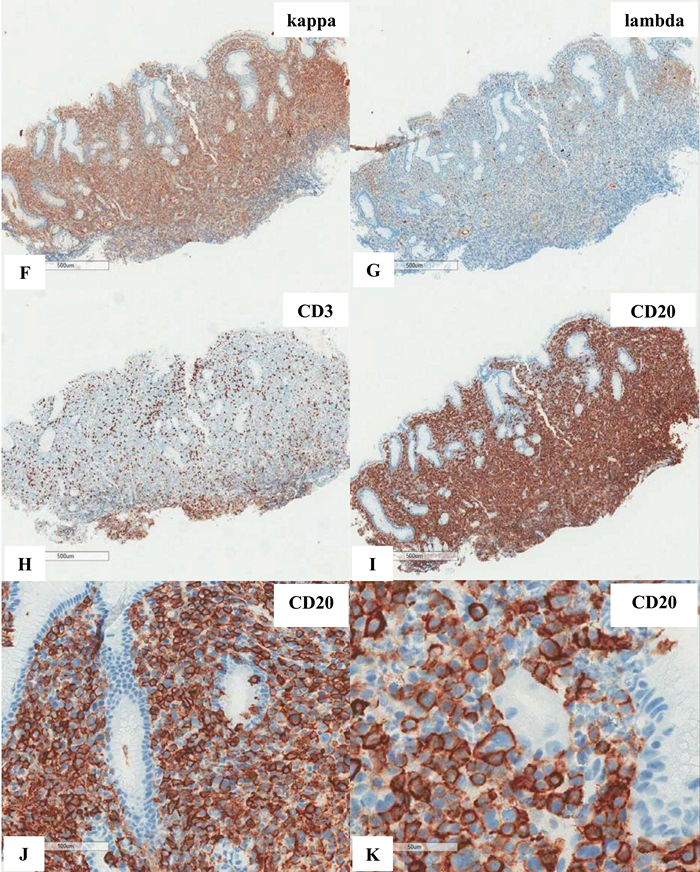
Figure 6
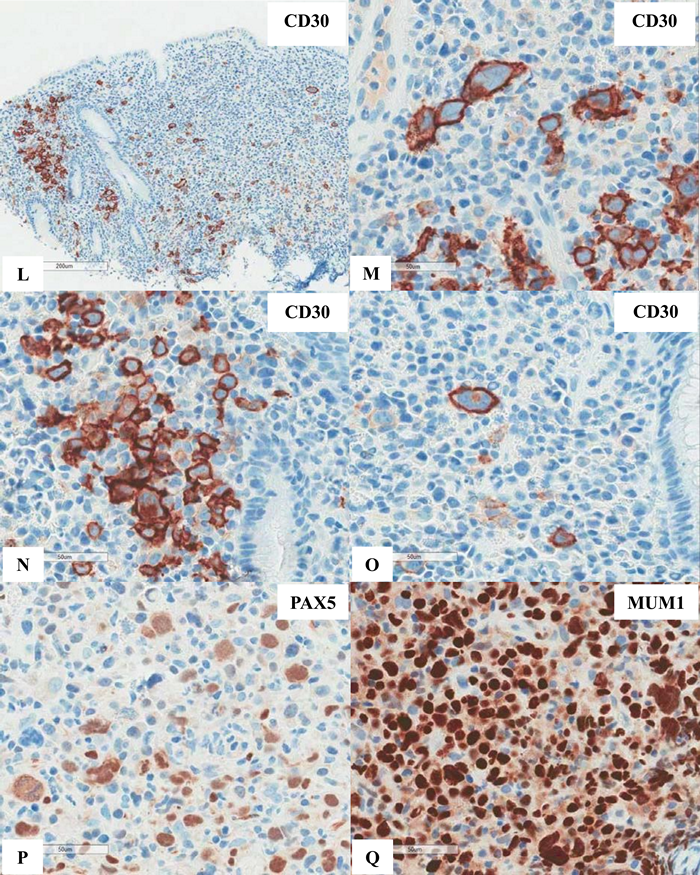
Figure 6
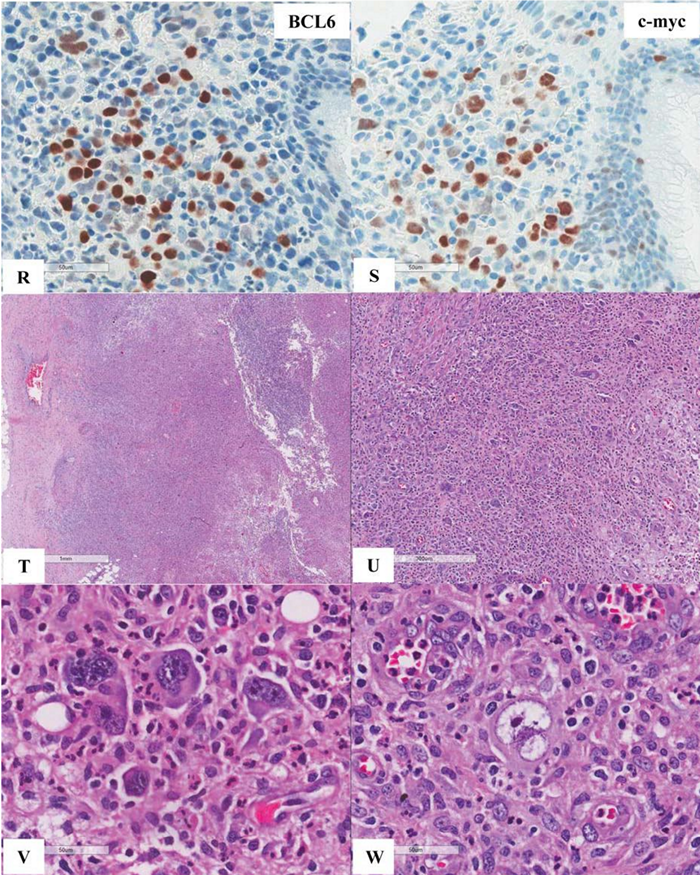
Figure 6
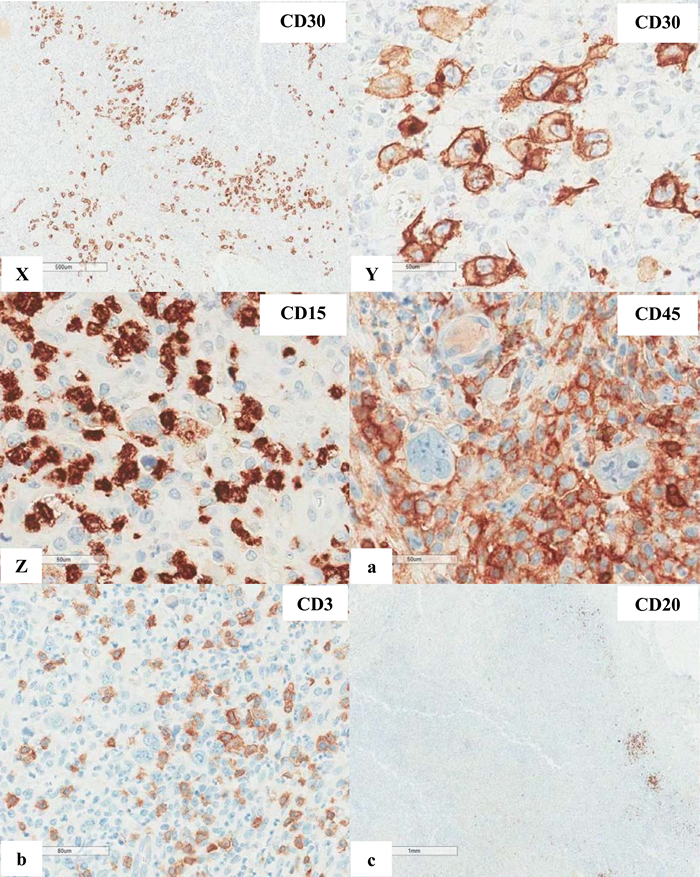
Figure 6
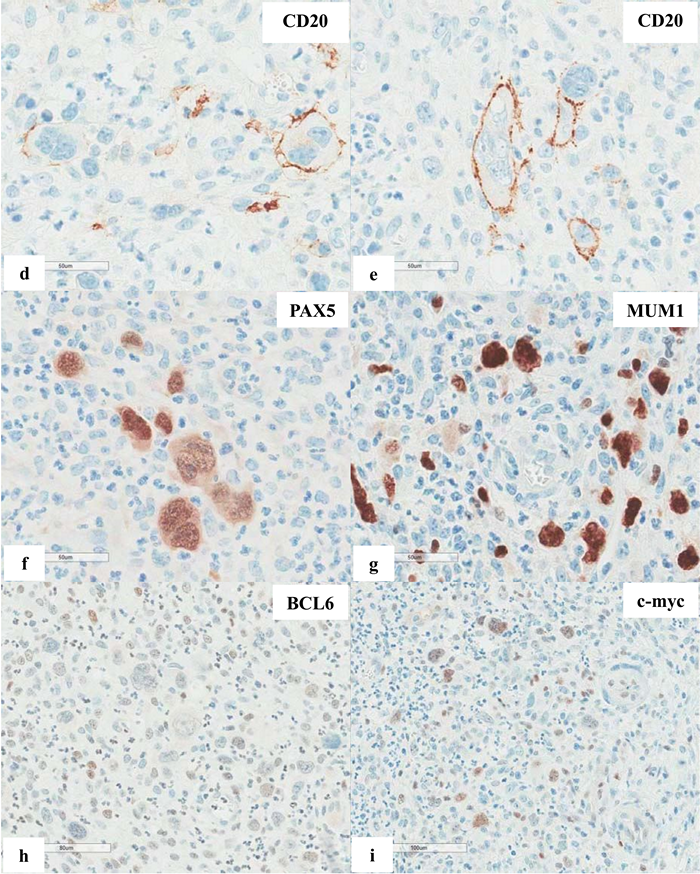
Figure 6 Extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue (MALT lymphoma) with occasional atypical large cells, some with binucleate or multinucleate forms. Occasional intracytoplasmic globules and intranuclear inclusions are also noted, suggestive of plasmacytic differentiation (PCD). F&G) Restriction to cytoplasmic kappa light chain, confirming PCD in this MALT lymphoma. H) A small number of CD3+ small reactive T-cells. I&J) CD20+ lymphoma cells. K) One CD20+ atypical large cell. L to O) CD30+ atypical large cells, varying in distribution. Some look like Reed-Sternberg cell. P) PAX5+ atypical large cells. Note decreased expression of PAX5 in typical lymphoma cells. Q) MUM1+ lymphoma cells including both typical and atypical ones. The MUM1+ small cells are plasma cells. R) Occasional BCL6+ large lymphoma cells, including occasional atypical ones. S) Expression of c-myc protein in occasional typical and atypical lymphoma cells. T) Low magnification of lymph node biopsy looks like nodular sclerosis classical Hodgkin lymphoma (CHL) with thick capsule. U) Occasional atypical large cells within the background of inflammatory cells, suggestive of CHL. V&W) Hodgkin-Reed-Sternberg cells at high magnification. X&Y) CD30+ lymphoma cells. Z) Two lymphoma cells with faint CD15 expression at the center. a) CD45- lymphoma cells. b) CD3- lymphoma cells. c) A small number of CD20+ lymphoma cells - not possible to appreciate at low magnification. d&e) CD20+ lymphoma cells. f to i) PAX5+, MUM1+, focal&faint BCL6+, and c-myc+ lymphoma cells - similar to those already seen in the first biopsy of MALT lymphoma, sufficient for diagnosis of Hodgkin-like large cell transformation.
2) Elastic transformation
Blastic transformation in this review emphasizes the morphologic transformation of the lymphoma cells. It is different from blastic transformation in chronic myelogenous leukemia whose stem cells progress to blasts either of myeloblastic or lymphoblastic type. It is also different from blastic phase in patients with malignant lymphoma when lymphoma
cells enter the circulation and they can be found in peripheral blood smears. When lymphoma patients receive combination chemotherapy and radiation, therapy-related myelodysplastic syndrome and secondary leukemia may occur. With the clinical information in mind, pathologists should consider second leukemia or leukemic infiltration when the biopsy obtained from the known case of lymphoma shows morphology different from the original lymphoma, especially with blastic or blastoid nuclear feature that is delicate nuclear chromatin pattern with or without conspicuous nucleolus/nucleoli. (Figure 7) Immunostaining to prove precursor or immature cell phenotype, including CD34 and TdT, will be helpful.
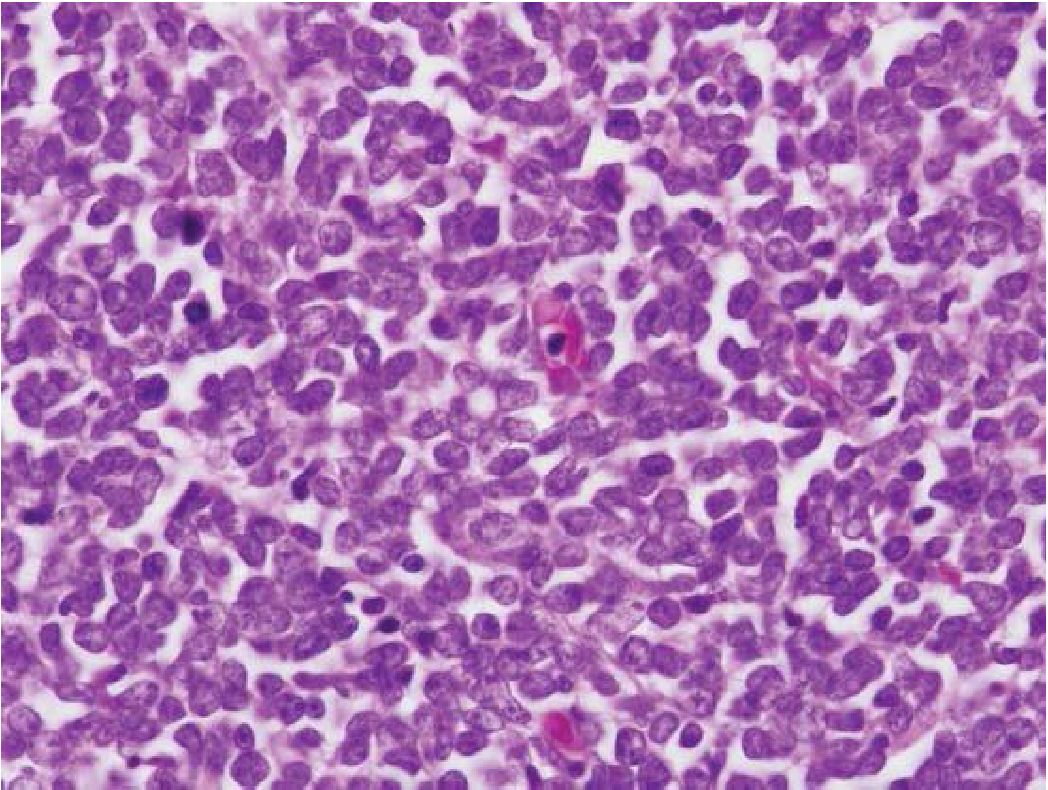
Figure 7 Blastic or blastoid nuclear feature. Note delicate nuclear chromatin and scant cytoplasm.
Blastic transformation is uncommon but it has been reported in FL and SLL/CLL. In FL, lymphoblast or blast transformation is preferred for report. But, in SLL/CLL, prolymphocytoid transformation is preferred as it is comparable to prolymphocytic leukemia3,7. Blastoid variant of mantle cell lymphoma (MCL) is another example that can be encountered with. Interestingly, both prolymphocytoid transformation in SLL/CLL and blastoid variant of MCL are not in a precursor stage as they do not express CD34 or TdT3.
3) Histiocytic transformation
Only a handful of cases that could demonstrate the relationship between the lymphoma cells and the neoplastic histiocytes. Again, these cases were reported in FL or SLL/CLL. The dedifferentiation of the lymphoma stem cells has been proposed like that applied to soft tissue sarcoma3. The difficulty to make a diagnosis of histiocytic transformation is how to exclude second neoplasm.
When histiocytic transformation does happen, the neoplastic histiocytes can be Langer-hans or non-Langerhans phenotype similar to that applied to histiocytic neoplasm. In daily pathology practice, distinction from hemophagocytic syndrome accompanying malignant lymphoma should be exercised according to nuclear pleomorphism, mitosis, increased proliferation index, etc. Reactive histiocytes can be found in any lesion so that the diagnosis of histiocytic neoplasm needs solid evidence3,20.
In summary, this review on morphologic approach for transformation in malignant lymphoma emphasizes the importance of morphologic recognition as well as salient features in clinical course of malignant lymphoma. A great deal of knowledge gathered from advance in immunophenotypic and genetic studies help us to understand malignant lymphoma better. A rising of new high throughput technologies will one day make this morphologic approach unnecessary for the diagnostic procedures as many biological markers involving in the pathogenesis of lymphoma will be used widely to control lymphoma growth or even to prevent lymphoma to occur. But, at this moment and several years to come, the morphologic approach is still the most cost effective way to provide the diagnosis of lymphoma and decision for further investigation. Nevertheless, diagnostic pitfalls can be encountered when immunophenotypic and genetic studies are employed so that pathologists should be familiar to the proper interpretation and learn more about limitation of any test investigated or misdiagnosis may occur21'23
REFERENCES
1. Murray JF, Agranat AL. Giant follicular lymphoblastoma with transformation to a reticulumcell sarcoma; a follow-up note. S Afr J Clin Sci. 1953;4:90-3.
2. Nowell PC. Phytohemagglutinin: an initiator of mitosis in cultures of normal human leukocytes. Cancer Res 1960;20:462-6.
3. Swerdlow SH, Campo E, Harris HL, Jaffe ES, Pileri SA, Stein H, et al, editors. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. Lyon (France): IARC; 2008.
4. Chigrinova E, Rinaldi A, Kwee I, Rossi D, Ran-coita PM, Strefford JC, et al. Two main genetic pathways lead to the transformation of chronic lymphocytic leukemia to Richter syndrome. Blood 2013;122:2673-82.
5. Jamroziak K, Tadmor T, Robak T, Polliack A. Richter syndrome in chronic lymphocytic leukemia: updates on biology, clinical features and therapy. Leuk Lymphoma 2015 Jan 21:1-10.
6. Strati P, Abruzzo LV, Wierda WG, O’Brien S,
Ferrajoli A, Keating MJ. Second Cancers and Richter Transformation Are the Leading Causes of Death in Patients With Trisomy 12 Chronic Lymphocytic Leukemia. Clin Lymphoma Myeloma Leuk 2015;15:420-7.
7. York JC, Glick AD, Cousar JB, Collins RD. Changes in the appearance of hematopoietic and lymphoid neoplasms: clinical, pathologic, and biologic implications. Hum Pathol 1984;15:11-38.
8. Oviatt DL, Cousar JB, Collins RD, Flexner JM, Stein RS. Malignant lymphomas of follicular center cell origin in humans. V. Incidence, clinical features, and prognostic implications of transformation of small cleaved cell nodular lymphoma. Cancer 1984;53:1109-14.
9. El-Galaly TC, Bilgrau AE, de Nully Brown P, Mylam KJ, Ahmad SA, Pedersen LM, et al. A population-based study of prognosis in advanced stage follicular lymphoma managed by watch and wait. Br J Haematol 2015;169:435-44.
10. Hirayama Y, Ishitani K, Ota S, Kurosawa M, Kondo T, Takimoto R, Mori A, et al. Long-term survey of survival time, histological transformation, and secondary malignancies in Japanese patients with advanced-stage follicular lymphoma in the rituximab era: Hokkaido Hematology Study Group. Int J Hematol 2014;100:281-9.
11. Bouska A, McKeithan TW, Deffenbacher KE, Lachel C, Wright GW, Iqbal J, et al. Genomewide copy-number analyses reveal genomic abnormalities involved in transformation of follicular lymphoma. Blood. 2014;123:1681-90.
12. Kridel R, Mottok A, Farinha P, Ben-Neriah S, Ennishi D, Zheng Y, et al. Cell-of-origin of transformed follicular lymphoma. Blood. 2015 Aug 25. pii: blood-2015-06-649905. [Epub ahead of print]
13. Isaacson PG. Gastrointestinal lymphoma. Hum Pathol. 1994;25:1020-9.
14. Greer JP, Salhany KE, Cousar JB, et al. Clinical features associated with transformation of cerebriform T-cell lymphoma to a large cell process. Hematol Oncol 1990;8:215-27.
15. Pulitzer M, Myskowski PL, Horwitz SM, Querfeld C, Connolly B, Li J, et al. Mycosis fungoides with large cell transformation: clin-icopathological features and prognostic factors. Pathology 2014;46:610-6.
16. Alberti-Violetti S, Talpur R, Schlichte M, Sui D, Duvic M. Advanced-stage mycosis fungoides and Sezary syndrome: survival and response to treatment. Clin Lymphoma Myeloma Leuk 2015;15:e105-12.
17. Kadin ME, Hughey LC, Wood GS. Large-cell transformation of mycosis fungoides-differ-ential diagnosis with implications for clinical management: a consensus statement of the US Cutaneous Lymphoma Consortium.[Commen-tary] J Am Acad Dermatol 2014;70:374-6.
18. Wehkamp U, Oschlies I, Nagel I, Brasch J, Kneba M, Gunther A, et al. ALK-positive primary cutaneous T-cell-lymphoma (CTCL) with unusual clinical presentation and aggressive course. J Cutan Pathol 2015 Jun 1. doi: 10.1111/ cup.12547. [Epub ahead of print]
19. Kuppers R, Engert A, Hansmann ML. Hodgkin lymphoma. J Clin Invest 2012;122:3439-47.
20. Stoecker MM, Wang E. Histiocytic/dendritic cell transformation of B-cell neoplasms: pathologic evidence of lineage conversion in differentiated hematolymphoid malignancies. Arch Pathol Lab Med 2013;137:865-70.
21. Sukpanichnant S. CD43 expression in malignant melanoma: a diagnostic pitfall. Asian Archives of Pathology 2014;10:97-109.
22. Sukpanichnant S, Pongpruttipan T. Commentary on the WHO Classification (2008) of Malignant Lymphoma. Siriraj Medical Journal 2009;61:154-9.
23. Pongpruttipan T, Sitthinamsuwan P, Rungkaew P, Ruangchira-urai R, Vongirad A, Sukpan-ichnant S. Pitfalls in classifying lymphomas. J Med Assoc Thai 2007;90:1129-36.


