Predictive Relevance of Tumor-infiltrating Lymphocytesin Breast Cancer
Tanin Titipungul1 M.D., Nipon Chaisuriya2 M.D., Emorn Phanomsri2M.Sc.,
Sakda Waraasawapati2 M.D., Supinda Koonmee2 M.D., Suwit Balthaisong2 B.Sc.,
Chawalit Pairojkul2 M.D., Yaovalux Chamgramol2 Ph.D.
1Department of Pathology, Mahasarakham Hospital, Mahasarakham, Thailand
2Department of Pathology, Faculty of Medicine, Khon Kaen University, Khon Kaen, Thailand
Correspondence: Yaovalux Chamgramol, Ph.D.
Department of Pathology, Faculty of Medicine, Khon Kaen University, Khon Kaen, 40002, Thailand.
Tel. +66-43-363691, Fax. +66-43-348388 E-mail: cyaova@yahoo.com
Received: 30 March 2014; Accepted 12 May 2014
ABSTRACT
Objective: To determine the prognostic significance of the immunophenotype, density and distribution of tumor-infiltrating lymphocytes (TILs) in breast cancer samples.
Materials and methods: This was a retrospective study, using paraffin-embedded samples obtained from 43 breast cancer patients, treated at Mahasarakham Hospital between 2012 and 2013. TILs and prognostic markers were evaluated by immunohistochemical staining of tissue microarray cores, employing monoclonal antibodies to lymphocyte markers (i.e., CD3, CD4, CD8 and CD20) and markers for breast cancer (i.e., ER, PR, HER2 and Ki-67). TILs were separated into 2 groups: 1) intratumoral lymphocytes (when found within tumor cell nests), and peritumoral lymphocytes (when infiltrating into the stroma, adjacent to the invasive front of tumor). The results of low- or high-density lymphocyte subtype infiltration of TILs in the intratumoral and peritumoral regions were compared with clinicopathological data.
Results: A positive correlation was found between high density peritumoral infiltrating CD8+ lymphocytes and breast carcinoma without lymph node metastasis (p=0.044), and an inverse correlation was found between the density of peritumoral infiltrated CD4+ lymphocytes and estrogen receptor expression (p=0.027). The density of peritumoral infiltrating CD8+ lymphocytes as well as the number of intratumoral CD20+ lymphocytes were independently, positively correlated with tumor size < 5 cm (p=0.027 and 0.004, respectively). Conclusion: Pathology assessment of TILs subtyping in breast cancer sections could have implications for patient prognosis.
Keywords: Breast cancer, tumor-infiltrating lymphocytes, CD3+, CD4+, CD8+, CD20+
INTRODUCTION
Tumor-infiltrating lymphocytes (TILs) arc lymphocytes that have infiltrated tumor tissue, both within the tumor itself (intratumoral lymphocytes, ITLs) or in the connective tissue surrounding the invasive margin of a tumor (peritumoral lymphocytes, PTLs)1 (Figure 1). TILs are considered to be a manifestation of the host anti-tumor response to alterations of tumor cells at the genomic and protcomic levels, associated with the acquisition of the neoplastic phenotype u-3. A complex cytokine and chcmokine milieu is present in the tumor microenvironment, allowing both anti- and pro-tumor immune responses. Such conflicting activities can often be detected within individual lesions45. While anti-tumor immune responses generally fail to control the growth of primary tumors, the quantity of infiltration by TILs is well-recognized as a favourable prognostic factor in various solid neoplasms6, including subtypes of breast cancer, with the strongest correlation for the latter observed in the ER-negative and HER2-positive subtypes7'9. Although there is no consensus on the individual immune cell subsets that consistently mediate this effect, an important link between pre-existing anti-tumor immune responses and long-tenn positive clinical outcomes has been established10. Breast cancer is one of the most common cancers among Thai women, which prompted us to investigate the prognostic value of TILs and immunophenotype the lymphocyte sub-populations using routine pathology methods on resected breast cancer specimens.
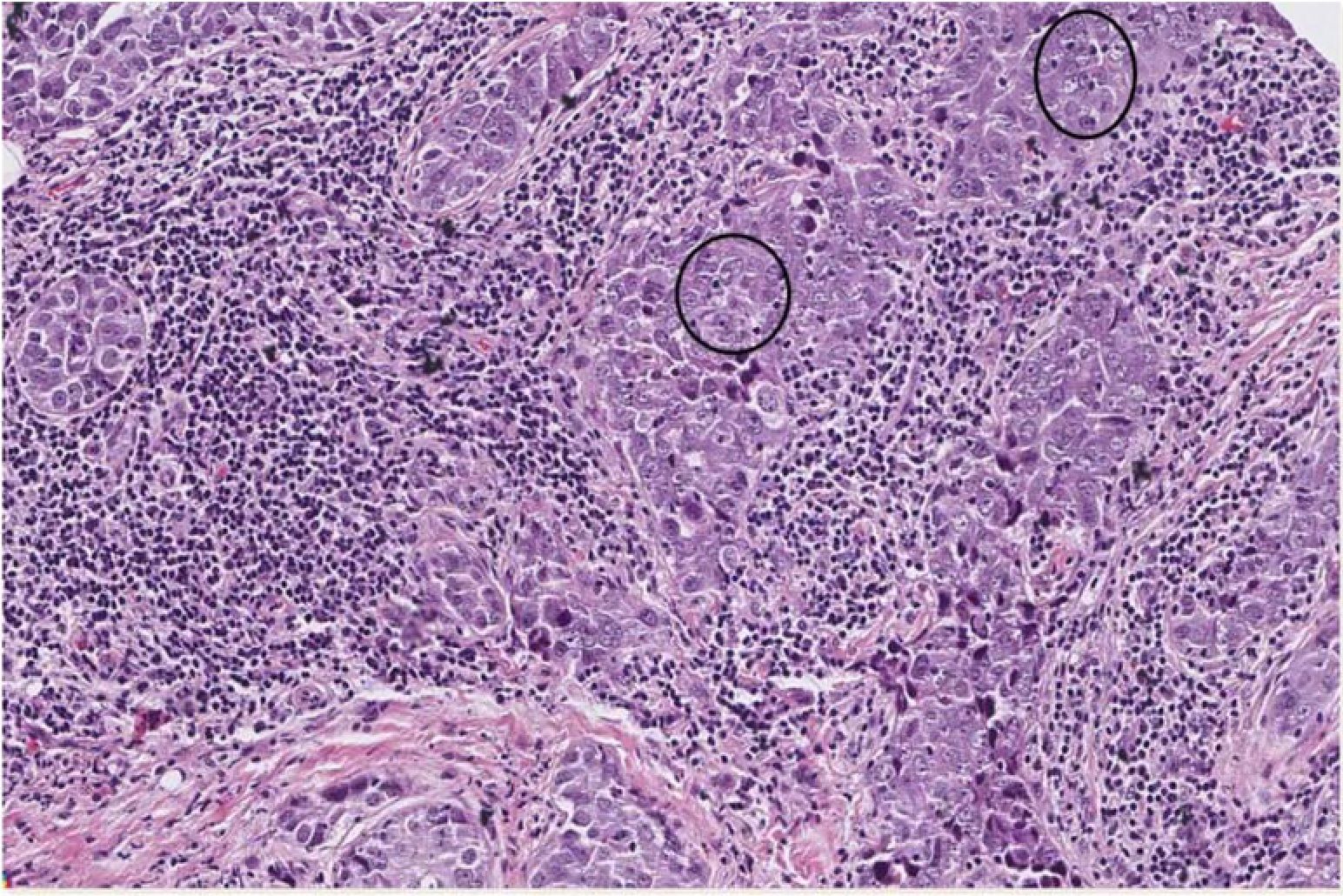
Figure 1 Micrograph of a breast cancer core, stained with H&E, showing tumor infiltrating lymphocytes (TILs) within an invasive ductal carcinoma (NOS). Peritumoral lymphocytes (PTLs) are seen extensively at the tumor-stroma boundaries and between the tumor sheets. Intratumoral lymphocytes (ITLs) are sparse but also evident in tumor nests (in circles). 200x magnification.
MATERIALS AND METHODS
Study samples :
The study samples were archival paraffin blocks obtained from 43 female breast carcinoma patients who were diagnosed and underwent mastectomy at Mahasarakham Hospital between 2012 and 2013. The patient files were reviewed to source the clinical and pathological data (including age, tumor size, tumor grade, axillary lymph node status, and lymphovascular invasion, staged according to the UICC, 7,h ed., 2009. None of the patients received preoperative chemotherapy, radiotherapy, or endocrine therapy. The protocols (ME 242556) used for human studies were approved by the Research Ethics Committee of Mahasarakham Hospital.
Tissue microarravs and immunohistochemistry :
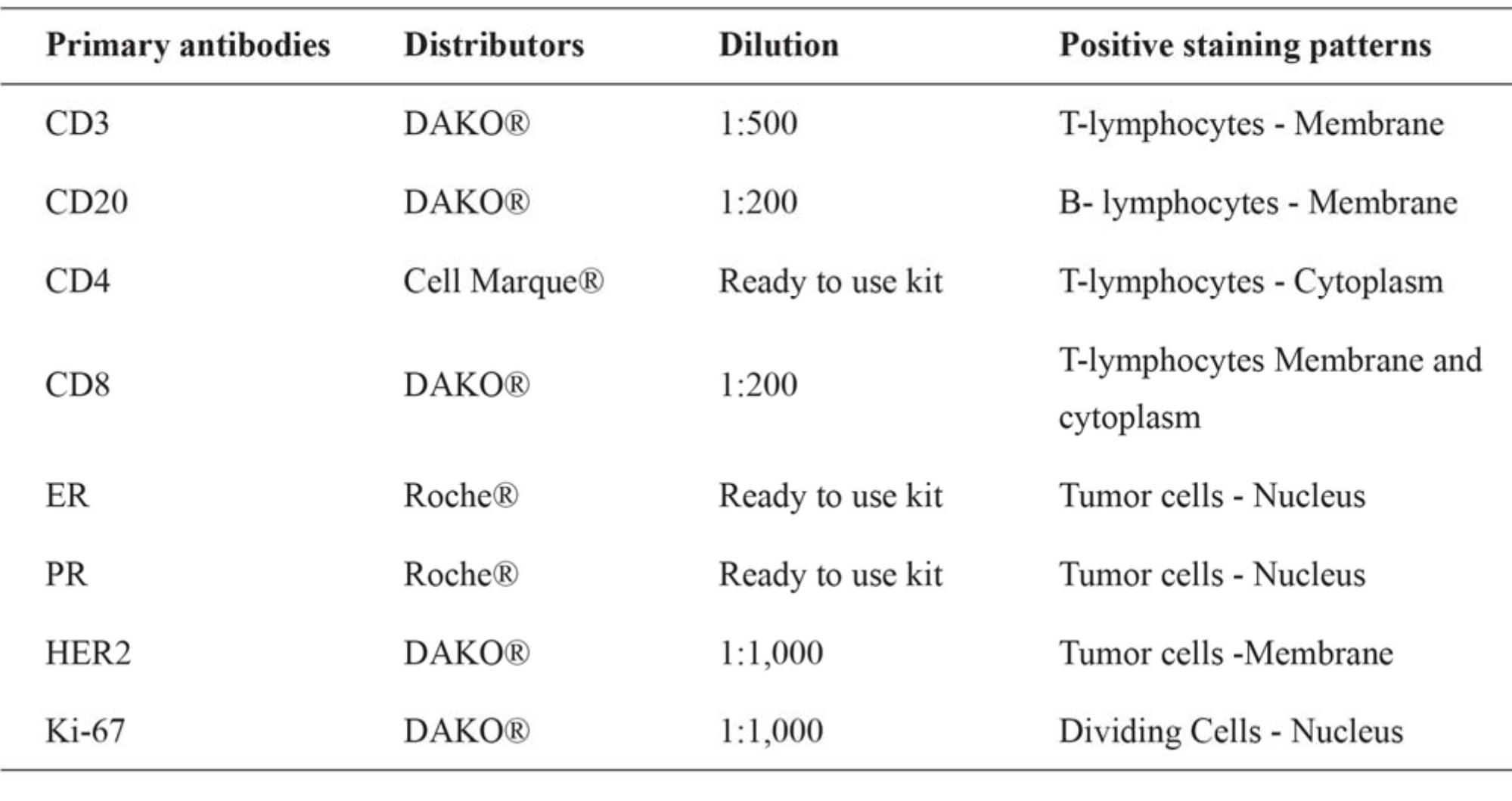
Breast cancer tissue microarrays (TMAs) with a 0.6-mm diameter x4 cores per case were prepared from tumor edges as previously described11. Freshly cut 4-micron sections from breast cancer TMAs were immunostained using the standard streptavidin-biotin complex method12. The antibodies and their sources, optimal dilution and interpretation sites are shown in Table 1. The mounted sections were deparaffinized with xylene, rehydrated in a graded scries of ethanol, soaked in 3% hydrogen-peroxide solution to quench endogenous peroxidase activity, and blocked with rabbit serum. Antigen retrieval was performed by microwave treatment in 10 mmol/L sodium citrate buffer (pH 6.0) for 20 minutes. After blocking the endogenous peroxidase, the sections were incubated in protein block solution. The sections were incubated overnight at 4°C with each of the primary antibodies. The sections were then treated with secondary antibodies conjugated to peroxidase-labeled polymer. Color development was performed using 3,3’-diaminobenzidine (DAB) and the sections were counterstaincd with haema-toxylin. The negative control was treated in parallel by omitting the primary antibody.
Table 1 Primary antibodies, dilution and positive staining patterns in this study.
Imniunohistochemical assay :
The immunohistochcmistry studies were interpreted by two pathologists (TP and SK), who were not apprised of the clinical data. Assessment of the full-face cores that showed areas of tumor and stroma were selected while necrotic areas were avoided. The density of the total lymphocytic infiltrate and the density of each cell immunophe-notype were grouped as: (a) intratumoral lymphocytes (ITLs) when found within tumor cell nests, and (b) peritumoral lymphocytes (PTLs) when adjacent stroma (at tumor cffacemcnt and vicinity) infiltration had occurred (Figure 1). The PTLs were semi-quan-titatively graded according to Kreike ct al., 2007[ 13] into: (a) absent, minimal (<10 lymphocytes/high-power field), (b) moderate (lymphocytes easily identified but no large aggregates); (Figure 2, B), and (c) extensive (large aggregates of lymphocytes in >50% of the tumor) (Figure 2, C and D). The ITLs were directly counted under X40 objective magnification. For statistical purposes, as suggested by West ct al., 20 ll14, ITLs were individually grouped as having a low or high number of lymphocyte infiltration, using the median value in the individual categories as cut-off points. PTLs were individually grouped as having a low density (grade 0 and 1+) or high density (grade 2+ and 3+) of lymphocyte infiltration. Expressions of ER, PR and Ki-67 in the nuclei were regarded as positive: (a) If >10% were positive for ER and PR, it was considered positive for the receptors, and (b) if >14% were positive for Ki-67, it was regarded as having a high labeling index. For HER2, its immunoreactivity was graded in each lesion according to ASCO-CAP guidelines, 2014: (a) negative (0, 1+), (b) equivocal (2+) and (c) positive (3+). In this study the negative and equivocal results were considered negative for HER2.
Statistical analysis :
All data were analyzed using IBM® SPSS version 19.0 (Khon Kaen University network license). The statistical tests for the correlation between clinicopathological parameters and lymphocyte infiltration within the tumor were the Chi-squared or Fisher’s exact test. We used the Fisher’s exact test when 20% of the expected frequencies were less than or equal to 5. Ap-valuc of <0.05 was considered statistically significant.
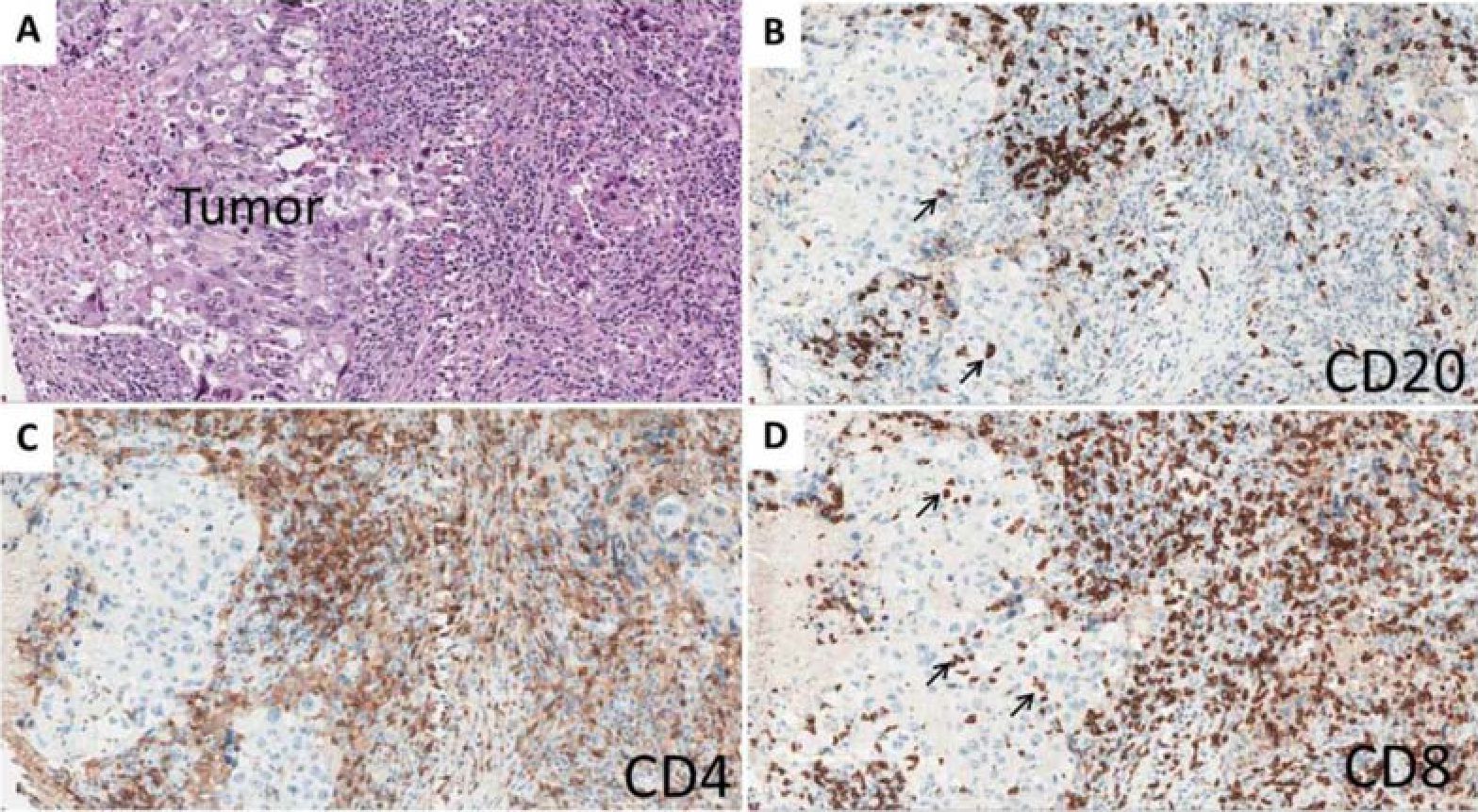
Figure 2 Micrographs of a representative breast cancer section. Tumor area labeled on H&E staining (A) and in serial cuts immunostained with lymphocyte markers, labeled at right lower comers (B-E, counterstained with haematoxylin). Co-localisation of lymphocyte subtypes in TILs are shown at various densities of lymphocyte infiltration in PTLs (A-D) and at ITLs (B and D, black arrows). 200x magnification.
RESULTS
Patient and tumor characteristics :
The clinicopathological characteristics of the 43 patients with invasive ductal carcinoma (NOS) of the breast are summarized in Table2.
Tumor-infiltrating lymphocytes in breast carcinoma :
Examination of tumors in H&E-stained sections revealed that TILs were evident in 42 of the 43 carcinomas (97.7%) while lymphocytes were rarely seen in the acinar epithelium of normal breast tissue in non-tumorous areas. Subacute inflammatory reactions were seen in the tumor necrotic foci. The infiltrating lymphocytes in the tumor-nests (ITLs) were sparse, but prominent in the tumor stroma at the edge of the tumor sheets and nearby stroma (PTLs), (Figure 1). Scoring of the density of PTLs showed that they were (a) absent in 1 of 43, (b) minimal (grade 1) in 8 of 43, (c) moderate (grade 2) in 15 of 43, and (d) extensive in 19 of 43 of carcinomas.
T and B lymphocytes :
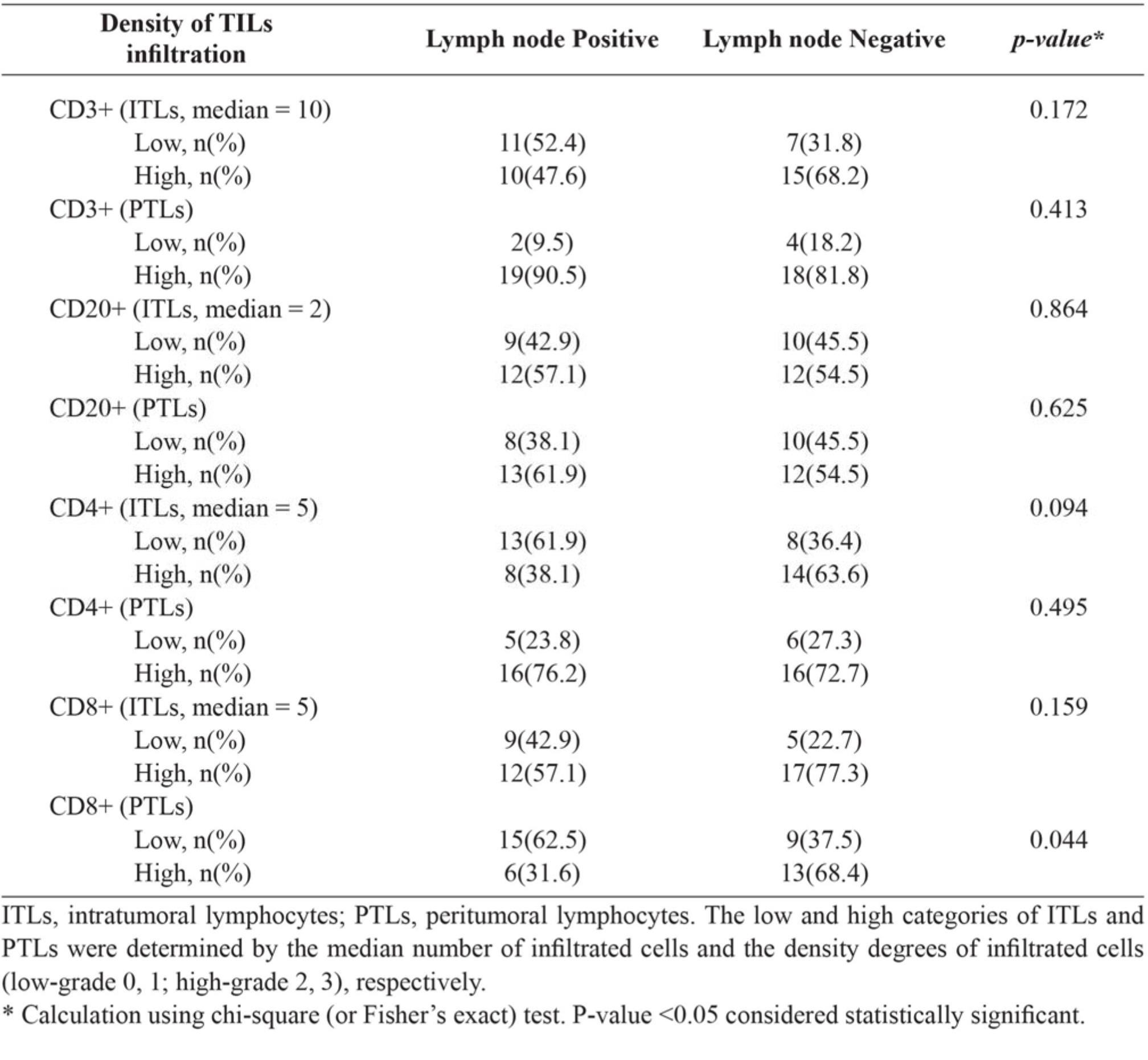
Immunophcnotyping demonstrated that the majority of cancer cases with TILs were CD3+ (T lymphocytes), which were present in 42 of 43 (97.7%) of the PTLs and 36 of 43 (83.7%) of the ITLs. Of the carcinoma cases, CD20+ B lymphocytes were found in 33 of 43 (76.7%) of the PTLs and 17 of 43 (39.5 %) of the ITLs. Co-localisation of T and B lymphocytes was evident in 33 of 42 (78.6%) carcinomas with PTLs and 26 of 36 (72.2%) carcinomas with ITLs. Notwithstanding, the correlation analysis of the density of T and B lymphocytes in ITLs and PTLs—compared to the clinicopathological variables—revealed that an association of ITLs with high CD20+ infiltration and tumor-size less than 5 cm (p=0.004) (Tables 2 and 3).
T lymphocyte subtyping :
The subpopulation of T lymphocytes in PTLs (n=42) included CD4+ T-hclpcr cells (41/42; 97.6%) and CD8+ cytotoxic T cells (37/42; 88.1%). In the ITLs, 32 of 36 (88.9%) and 27 of 36 (75%) were CD8+ and CD4+, respectively. The respective correlation of the degree of CD4+ and CD8+ proliferation in the ITLs and PTLs compared to the clinicopathological parameters was significant: viz., (a) carcinomas with negative estrogen receptors correlated with a high density of CD4+ lymphocytes in the PTLs (p=0.027) (Table 4) and (b) carcinoma with tumor size less than 5 cm is correlated with high density ofCD8+ lymphocytes in the PTLs (p=().()27) (Table 5), and (c) a positive correlation was found between high density pcritumoral infiltrated CD8+ lymphocytes and breast carcinoma without lymph node metastasis (p=0.044) (Table 6).
Table 2 Correlation lymphocyte-subtype densities, and infiltrating locations (ITLs or PTLs) of tumor-infiltrating lymphocytes (TILs) with clinicopathological valiables in 43 breast cancers.
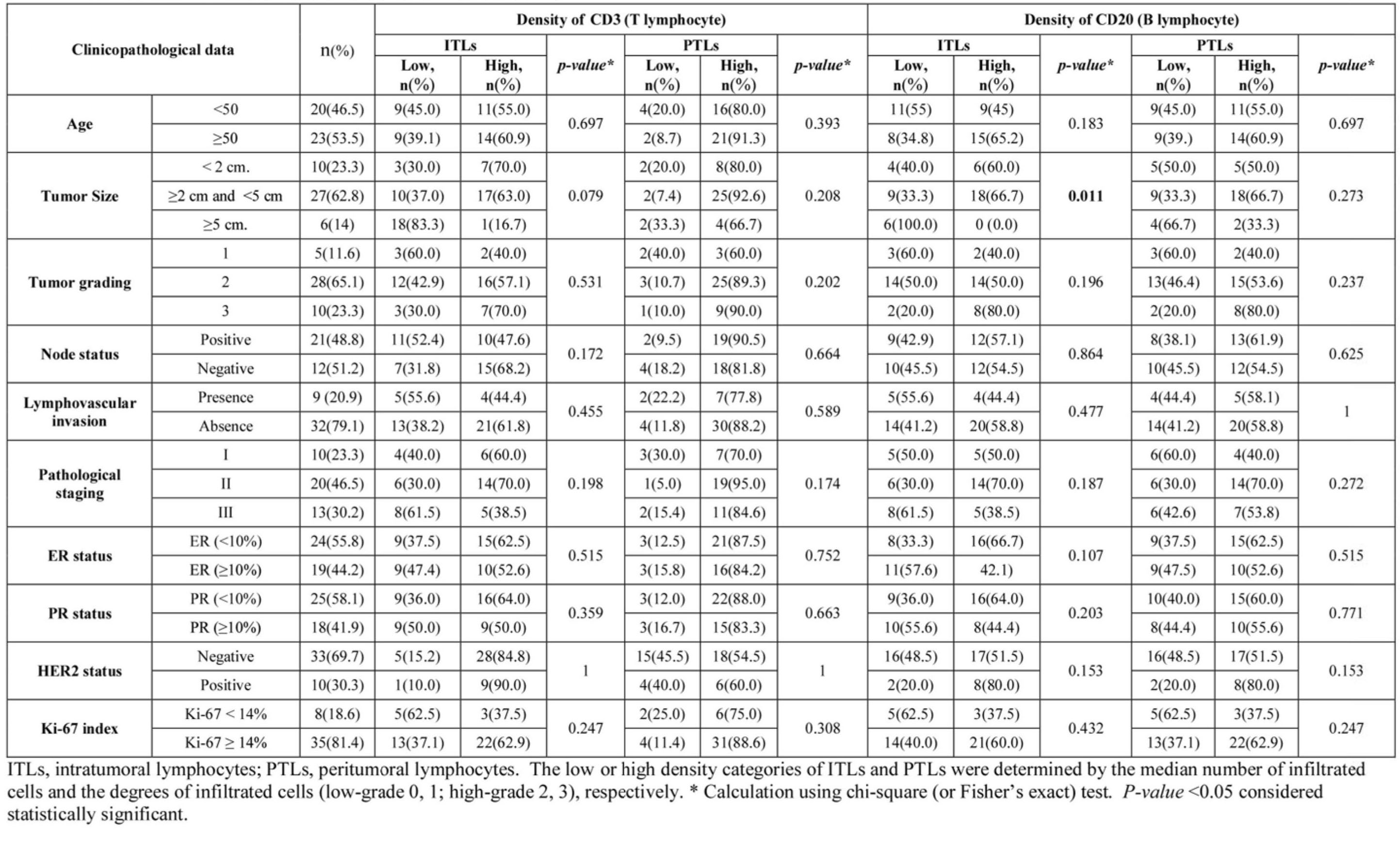
Table 3 Correlation between tumor sizes and density of CD20+ lymphocytes at ITLs and PTLs.
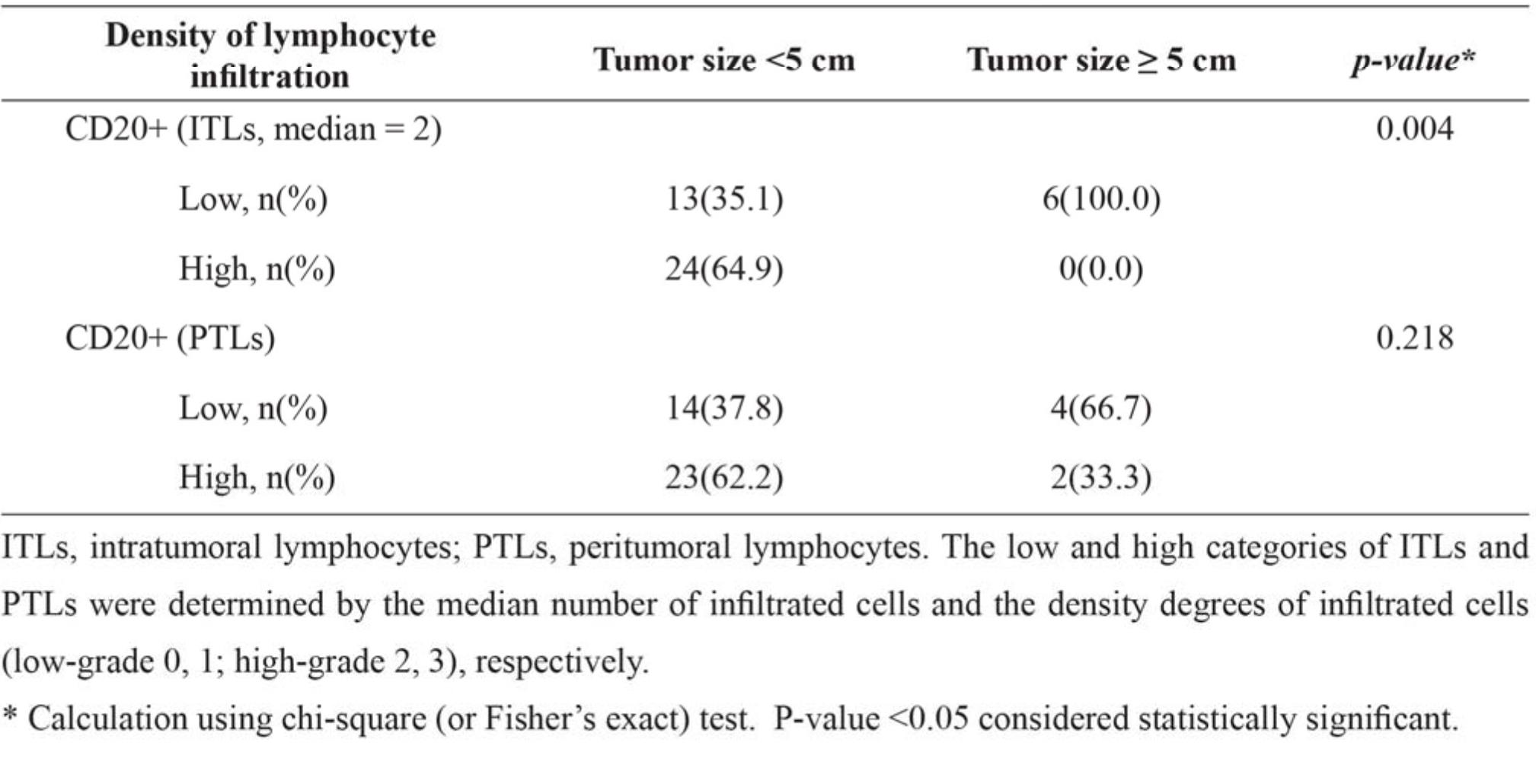
Table 4 Correlation between estrogen receptor status and lymphocyte-subtype densities, infiltrating locations (ITLs or PTLs) of tumor-infiltrating lymphocytes (TILs).
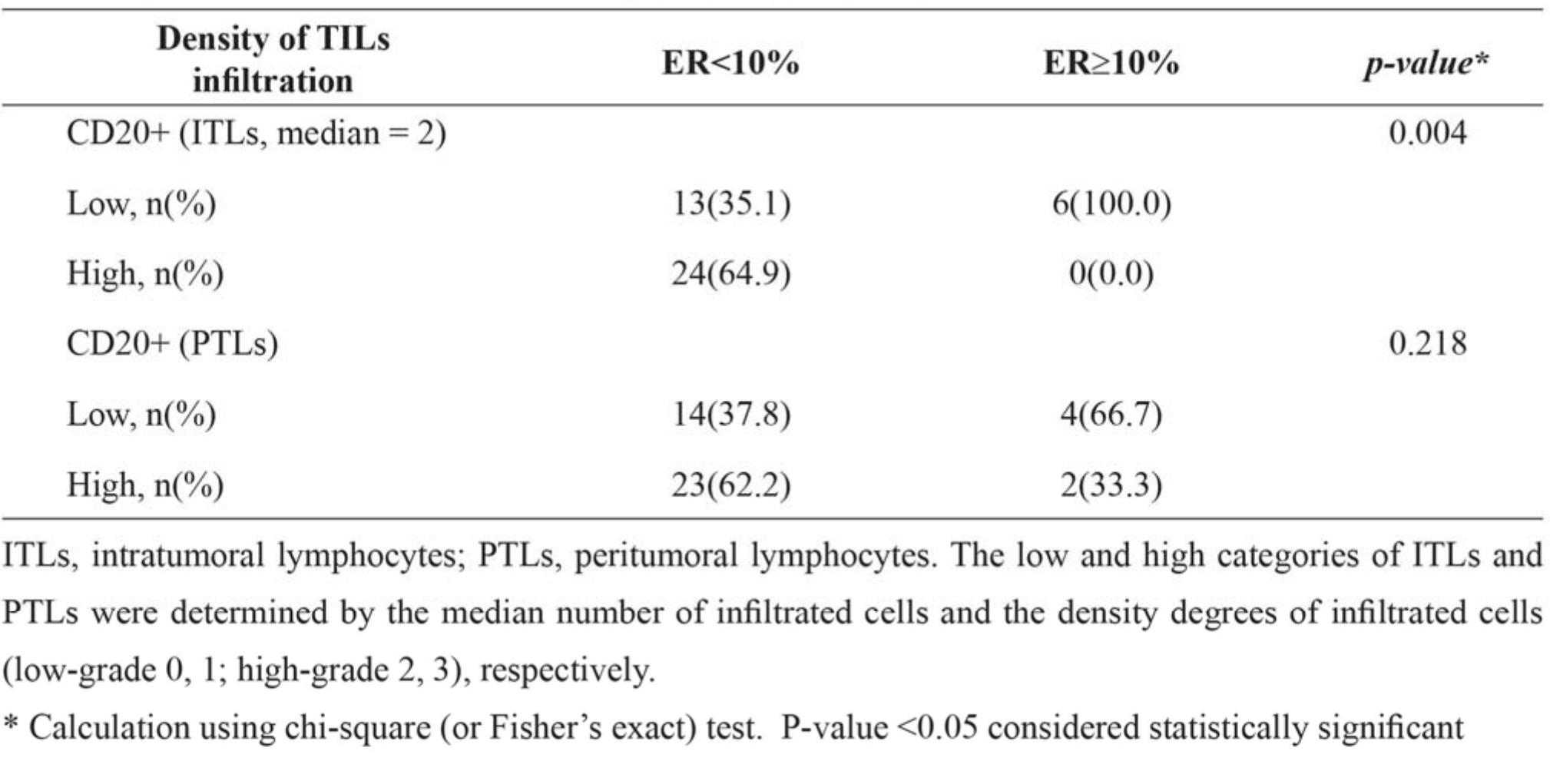
Table 5 Correlation between tumor sizes and density of CD8+ lymphocytes at different locations (ITLs or PTLs) of tumor-infiltrating lymphocytes (TILs).
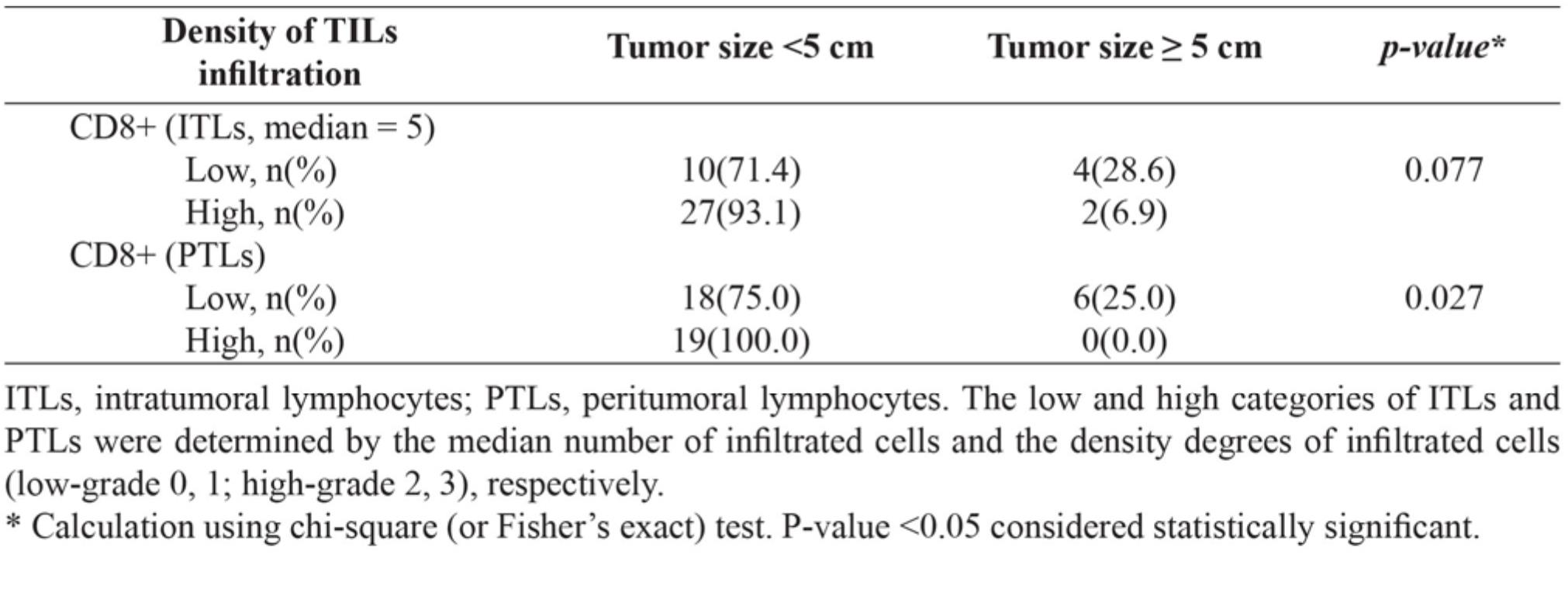
Table 6 Correlation between lymph node status and lymphocyte-subtype densities, infiltrating locations (ITLs or PTLs) of tumor-infiltrating lymphocytes (TILs).
DISCUSSION
In the current study, we observed a high frequency of TILs in invasive breast carcinomas. The presence of lymphocyte infiltration in tumors is consistent with previous reports comparing TILs in benign breast lesions, DCIS and lobular carcinoma15- l6. The presence of these lymphocytes is, moreover, considered an expression of an antitumor immune response, capable of preventing progression and metastasis1. In cancer progression, the key factors for recruitment of TILs are soluble factors (i.c., proteases, growth factors, cytokines and chemokines) locally-secreted by stromal and tumor cells1718. Our findings suggest that T and B lymphocytes were co-recruited in 72.2% and 75.8% of ITLs and PTLs, respectively. This could be evidence of chemokine mediation as well as a local inflammatory reaction18. ITLs principally comprised of CD4+ and CD8+ T cells, and B cells2 (Figure 2, B-D). CD4+ T cells perform critical roles in recruiting, activating, and regulating many facets of the adaptive immune response; with their helper functions for B cells and CD8+ cytotoxic T cell-mediated responses being well-documented1-2. Our observation of a high density of peritumoral infiltrated CD8+ lymphocytes as well as the number of intratumoral CD20+ lymphocytes were independently, positively correlated with tumor size < 5 cm (p=0.027 and 0.004, respectively). CD8+ cytotoxic T cells are considered a key component of an effective antitumor immunity2; so the observation of an increase in CD8+ cytotoxic T cells only at a certain size likely therefore represents the balance between anti- and pro-tumor immune responses, which fail to control the growth of primary tumors2. Similarly, but independently observed, are the increasing number of B lymphocytes at ITLs, indicating that TILs generate memory cells of intratumoral CD20+ lymphocytes that mediate anticancer immunosurveillance19 which also fails to eliminate malignant cells at later stages of tumorigenesis20. Another example when B lymphocyte infiltration fails to contain breast cancer growth is medullary breast carcinoma: a rare subtype of high-grade invasive ductal carcinoma characterized by dense infiltration of B lymphocytes and plasma cells both around and within the tumor. Earlier studies suggested that the lymphocytic response may be the key factor associated with a better prognosis over against conventional ductal carcinomas21. In a study done after re-defining the histological criteria of 104 cases of medullary carcinoma (a series of 1,411 breast cancers with similar staging), deaths due to the carcinoma were reportedly rare after 5 years, while the unsuccessfully-treated medullary carcinoma patients died fastest of all groups22. The authors concluded that medullary carcinomas are essentially aggressive malignancies, as indicated by their tumor grade, but that their biological potential is countered to a considerable extent by the host’s immune response22. Importantly, a recent gene expression profiling study linked a poorer prognosis among the subset of medullary breast carcinomas carrying the BRCA1 mutation23-24. Studies of human breast carcinomas indicate the immunogenic intrinsic nature of the tumor, as several autoantigens have been identified (i.e., the HER2/neu protein, p53, CEA, c-Hras, c-myc and MUC-1)25. We observed that a high density peritumoral infiltrated CD4+ lymphocytes was inversely correlated with estrogen receptor expression (p=0.027), linking TILs to the intrinsic properties of the cancer cells. Estrogen is a steroid hormone involved in regulating the differentiation and proliferation of breast epithelial cells. Estrogen influences cells by interacting with the ER in the nucleus, eliciting a cascade of transcriptional regulatory activity directly modulating the expression of cell-cycle regulatory and growth factor receptor pathways26. It is well-recognized that ER-positive and ER-negative breast cancers are two different disease entities27. Generally, ER-negative tumors tend to be of a high grade, are more frequently p53 mutation, and have a worse prognosis than ER-positive disease27. A high immune signal has been linked with improved patient outcomes in subtypes of breast cancers that are ER-negative and HER2-positive7 9.
One of the most important independent prognostic factors in breast carcinoma is evidence of axillary lymph node metastasis. Laguens ct al. (2012) identified and quantified CD4+ and CD8+ T cells in the stroma of human breast cancer and correlated them with the presence of CXCL928. The CXCL9 is a monokine induced by interferon gamma (MIG) that targets lymphocytes17 28. They demonstrated that the number of CD4+ and CD8+ T cells in breast cancer tissue was significantly increased with a clear predominance of CD8+ T cells, while MIG/CXCL9 levels were significantly elevated with respect to normal breast tissue28. Actually, this chemokine correlated with the number of CD8+ T cells only in non-metastatic tumors28. These data suggest that MIG targets cytotoxic T cells and can play a critical role in malignant progression. Evidence in support of their hypothesis is found in our observation of a positive correlation between high-density peritumoral infiltration of CD8+ lymphocytes and breast carcinoma without lymph node metastasis (p=0.044). The underlying mechanism is possibly inhibition of micro-metastasis29. We observed a histopathological association between the immune subtypes of TILs at the tumor edges and have shown how these are implicated in tumor prognosis. The study was limited by the small number of available samples, and the options for an appropriate statistical analysis. The TILs count represent continuous variables (ITLs), and the PTLs count were ordinal outcomes. We therefore have to converted the continuous ITLs data and ordinal PTLs data into categorical data before applying a univariate analysis and nonparametric testing (Chi-square or Fisher exact). The data modification may have weakened the correlation analysis, i.e., after trying repeated multiple comparisons of tumor size, grade and stage with continuous variable (ITLs) using an ANOVA and post-hoc test for Bonferroni correction, a significant association was only found for B-cells with tumor size (but not T-cells). More cases plus functional studies are needed, then an analysis of the relationship between the density and specific subtypes of TILs can be tested and the correlation determined between the manifestation of a host anti-tumor reaction and predictive tumor grading and staging.
CONCLUSION
We provided confirmation that TILs are a common manifestation in ductal breast cancer30 and that breast cancer is immunogenic for recruitment of T and B lymphocytes. The individual immuno-sub-types of TILs may be a further prognostic indicator of use in routine pathological staging of the tumor.
ACKNOWLEDGEMENTS
We thank the Department of Pathology and the Faculty of Medicine for support and Mr. Bryan Roderick Hamman for assistance with the English.
REFERENCES
1. Chiou SH, Sheu BC, Chang WC, ct al. Current concepts of tumor-infiltrating lymphocytes in human malignancies. J Reprod Immunol 2005 Oct; 67(1-2): 35-50.
2. Menegaz RA, Michclin MA, Etchcbehcre RM, Fernandes PCJ, Murta EFC. Peri- and intratu-moral T and B lymphocytic infiltration in breast cancer. Eur J Gynaecol Oncol 2008; 29(4): 321-6.
3. Simonson WTN, Allison KH. Tumor-infiltrating lymphocytes in cancer: Implications for the diagnostic pathologist. Diagnostic Histopathology 2011; 17(2): 80-90.
4. Scurr M, Gallimore A, Godkin A. T cell subsets and colorectal cancer: Discerning the good from the bad. Cell Immunol 2012; 279: 21-4.
5. DeNardo DG, Andreu P, Coussens LM. Interactions between lymphocytes and myeloid cells regulate pro-versus anti-tumor immunity. Cancer Metastasis Rev 2010; 29: 309-16.
6. Jochems C, Schlom J. Tumor-infiltrating immune cells and prognosis: The potential link between conventional cancer therapy and immunity. Exp Biol Med (Maywood) 2011; 236: 567-79.
7. Teschendorff AE, Miremadi A, Pinder SE, Ellis IO, Caldas C. An immune response gene expression module identifies a good prognosis subtype in estrogen receptor negative breast cancer. Genome Biol 2007; 8(8): R157.
8. Alexe G, Dalgin GS, Scanfcld D, ct al. High expression of lymphocyte-associated genes in node-negative HER2+ breast cancers correlates with lower recurrence rates. Cancer Res 2007; 67(22): 10669-76.
9. Desmcdt C, Haibc-Kains B, Wirapati P, et al. Biological processes associated with breast cancer clinical outcome depend on the molecular subtypes. Clin Cancer Res 2008; 14(16):5158-65.
10. Rahir G, Moser M. Tumor microenvironment and lymphocyte infiltration. Cancer Immunol Immunother 2012; 61(6): 751-9.
11. Camp RL, Charette LA, Rimm DL. Validation of tissue microarray technology in breast carcinoma. Lab Invest 2000; 80: 1943-9.
12. Hsu S, Raine L. Protein, avidin and biotin in immunohistochcmistry. J Histochcm Cytochcm 1981; 29: 1349-53.
13. Kreike B, Van Kouwcnhove M, Horlings H, et al. Gene expression profiling and histopatho-logical characterization of triple-ncgativc/basal like breast carcinoma. Breast Cancer Res 2007; 9: R65.
14. West NR, Milne K, Truong PT, et al. Tumor-infiltrating lymphocytes predict response to anthracycline-based chemotherapy in estrogen receptor-negative breast cancer. Breast Cancer Res BCR 2011; 13(6): R126.
15. Bcn-Hur H, Cohen O, Schneider D. The role of lymphocytes and macrophages in human breast tumorigencsis: An immunohistochcmical and morphometric study. Anticancer Res 2002; 22: 1231-8.
16. Hussein MR, Hassan HI. Analysis of the mononuclear inflammatory cell infiltrate in the normal breast, benign proliferative breast disease, in situ and infiltrating ductal breast carcinomas: Preliminary observations. J Clin Pathol 2006; 59: 972-7.
17. Tanaka T, Bai Z, Srinoulprasert Y, Yang BG, Hayasaka H and Miyasaka M. Chcmokines in Tumor Progression and Metastasis. Cancer Science 2008; 96(6): 317-22.
18. Raman D, Baugher PJ, Thu YM and Richmond A. Role of Chemokines in Tumor Growth. Cancer Letters 2007; 256(2): 137-65.
19. Gu-Tranticn C, Willard-Gallo K. Tumor-infiltrating follicular helper T cells: The new kids on the block. Oncoimmunology 2013 Oct 1; 2(10): e26066. Epub 2013 Sep 12.
20. De Nardo DG and Coussens LM. Balancing Immune Response: Crosstalk between Adaptative and Innate Immune Cells during Breast Cancer Progression. Breast Cancer Research 2007; 9(4): 212-22.
21. Moore OS, Foote FW. The relatively favourable prognosis of medullary carcinoma of the breast. Cancer 1949; 2: 635-42.
22. Bloom HJG, Richardson WW, Field JR. Host resistance and survival in carcinoma of breast: A study of 104 cases of medullary carcinoma in a scries of 1411 cases of breast cancer followed for 20 years. Br Med J 1970; 3: 181-8.
23. Jacquemicr J, Padovani L, Rabayrol L, et al. Typical medullary breast carcinomas have a basal/myocpithelial phenotype. J Pathol 2005; 207: 260-8.
24. Stoppa-Lyonnet D, Ansquer Y, Dreyfus H, et al. Familial invasive breast cancers: Worse outcome related to BRCA1 mutations. J Clin Oncol 2000; 18:4053-9.
25. Nzula S, Going JJ, Stott DI. The role of B lymphocytes in breast cancer: A review and current status. Cancer Therapy 2003; 1: 353-62.
26. Dickson RB and Lippman ME. Estrogenic regulation of growth and polypeptide growth factor secretion in human breast carcinoma Endocr Rev 1987; 8: 29.
27. van't Veer LJ, Dai H, van dc Vijver MJ, et al. Gene expression profiling predicts clinical outcome of breast cancer. Nature 2002; 415: 530-6.
28. Laguens G, Coronato S, Chambo J and Girolamo V. Stromal CD4+ and CD8+ T Cells in Human Breast Carcinomas. Its Correlation with Chcmokinc MIG/CXCL9. Advances in Breast Cancer Research 2012; 1:7-11
29. Giraldo NA, Bccht E, Remark R, et al. The immune contexture of primary and metastatic human tumors. Curr Opin Immunol 2014 Jan 30; 27C: 8-15.
30. Colotta F, Allavena P, Sica A, et al. Cancer-related inflammation, the seventh hallmark of cancer: Links to genetic instability. Carcinogenesis 2009; 30: 1073-81.


