Comparison of a-smooth muscle actin expression between squamous cell carcinoma and pseudoepitheliomatous hyperplasia
Juthamas Ngamwiseschaikul, MD1, Juthamas Wongphum, BSc1, Somboon Keelawat, MD1
Departments of Pathology, Faculty of Medicine, Chulalongkorn University, Bangkok, Thailand
Corresponding author: Somboon Keelawat, M.D.
Department of Pathology, Faculty of Medicine, Chulalongkorn University,
Pathumwan, Bangkok, Thailand 10330
Tel: 662-256-4235 Fax: 662-652-4208 Email: trcskl@gmail.com
Received: 6 April 15; Accepted 5 June 15
ABSTRACT
Objective: To evaluate the usefulness of a-smooth muscle actin (a-SMA) expression of reactive myofibro-blastic cells within desmoplastic reaction tissue for distinction between invasive squamous cell carcinoma (SCC) and pseudoepitheliomatous hyperplasia (PEH).
Materials and Methods: Twenty-one cases of SCC and 20 cases of PEH were immunologically stained with a-SMA antibody. The immunohistochemical staining is evaluated and recorded in percentage of staining area in stromal tissue per the total stroma obtained in each slide (excluding blood vessels’ wall, muscle, inflammatory and reparative tissue areas).
Results: The average percentage of positive staining is significantly higher in the reactive stroma of SCC compared to the stroma of PEH (18.57% VS 1.75%) (p=0.014). With the cut off value at 10% staining, the sensitivity of staining for diagnosis of SCC is 52.38%, specificity 95%, PPV 91.67%, NPV 65.52%. Conclusion: The a-SMA staining is highly specific for detection of desmoplastic reaction in SCC. However, the weakness of this marker is its low sensitivity.
Running title: Alpha smooth muscle actin expression of squamous cell carcinoma and pseudoepitheliomatous hyperplasia
Keywords: Alpha smooth muscle actin, Squamous cell carcinoma and Pseudoepitheliomatous hyperplasia
INTRODUCTION
One of the difficulties in routine practice for pathologists occurs while dealing with the diagnosis of SCC based on small biopsied specimens. There are several conditions that can be confused with invasive carcinomas such as pseudoepitheliomatous hyperplasia, squamous proliferation with poor tissue orientation and necrotizing sialometaplasia. Oftentimes, decisions between whether the submitted lesions are benign or malignant cannot be made due to equivocality of the morphology which can result in delay of treatment.
Several studies have tried to overcome these limitations by utilizing immunohistochemical stains. Studies on p53, matrix metalloproteinase 1 (MMP-1) and E-cadherin stains have, to some extent, shown usefulness in differentiating invasive squamous cell carcinoma from pseudoepitheliomatous hyperplasia1. Alpha SMA which is an excellent marker for myofibroblasts has also been described to be beneficial in separation of SCC from benign mucosa2-3
However, to the authors’ knowledge, there has been no study of a-SMA on PEH in comparison to SCC. But, since PEH is one lesion that can cause diagnostic dilemmas with carcinomas and a-SMA has been claimed to be useful in distinguishing SCC from several benign lesions, therefore, in this study, the authors aim to assess and compare immunoex-pression of a-SMA between the reactive stromal cells around the invasive squamous cell carcinoma and pseudoepitheliomatous hyperplasia with an attempt to separate these two lesions by this marker.
MATERIAL AND METHOD
This study was carried out at the Department of Pathology, Faculty of Medicine, Chulalongkom University, Bangkok, Thailand.
Recruited samples
Samples included 21 cases of squamous cell carcinoma and 20 cases of pseudoepitheliomatous hyperplasia froml999 to 2011. These cases were selected from lesions of various sites. The H&E sections of all selected cases were reviewed to confirm the diagnoses.
Immunohistochemistry
Immunohistochemistry was performed on paraffin-embedded sections of all specimens using primary antibody specific to a-SMA (Dako, USA). Two-micron sections were placed on positively charged slides (Platinum Pro, Matsunami, Japan). Thereafter, the sections were further proceeded for antigen retrieval and immunostaining by standard protocol of the Ventana automated immunostainer (Ventana Medical Systems Inc., USA).
Semiquantitative assessment of a-SMA expression
All a-SMA antibody stained slides were examined under the light microscope (figure 1 B, D). Blood vessels present within the connective tissue of the immunostained sections served as positive internal control (figure ID). Spindle cells in the stroma which are positive for a -SMA were regarded as myofibroblasts if an intracytoplasmic brown color staining was present regardless of its intensity (figure IB).
The evaluation of a-SM Aexpression was done at the invasive front of squamous cell carcinoma and the stroma close to the reactive pseudoepitheliomatous epithelium. Immunostaining was assessed by the evaluation of percentage of area with a-SMA expression per the total stroma obtained in each slide (excluding blood vessels’ wall, muscle, inflammatory and reparative tissue areas). The scoring of a -SMA stain for each case was determined by two authors (JN and SK).
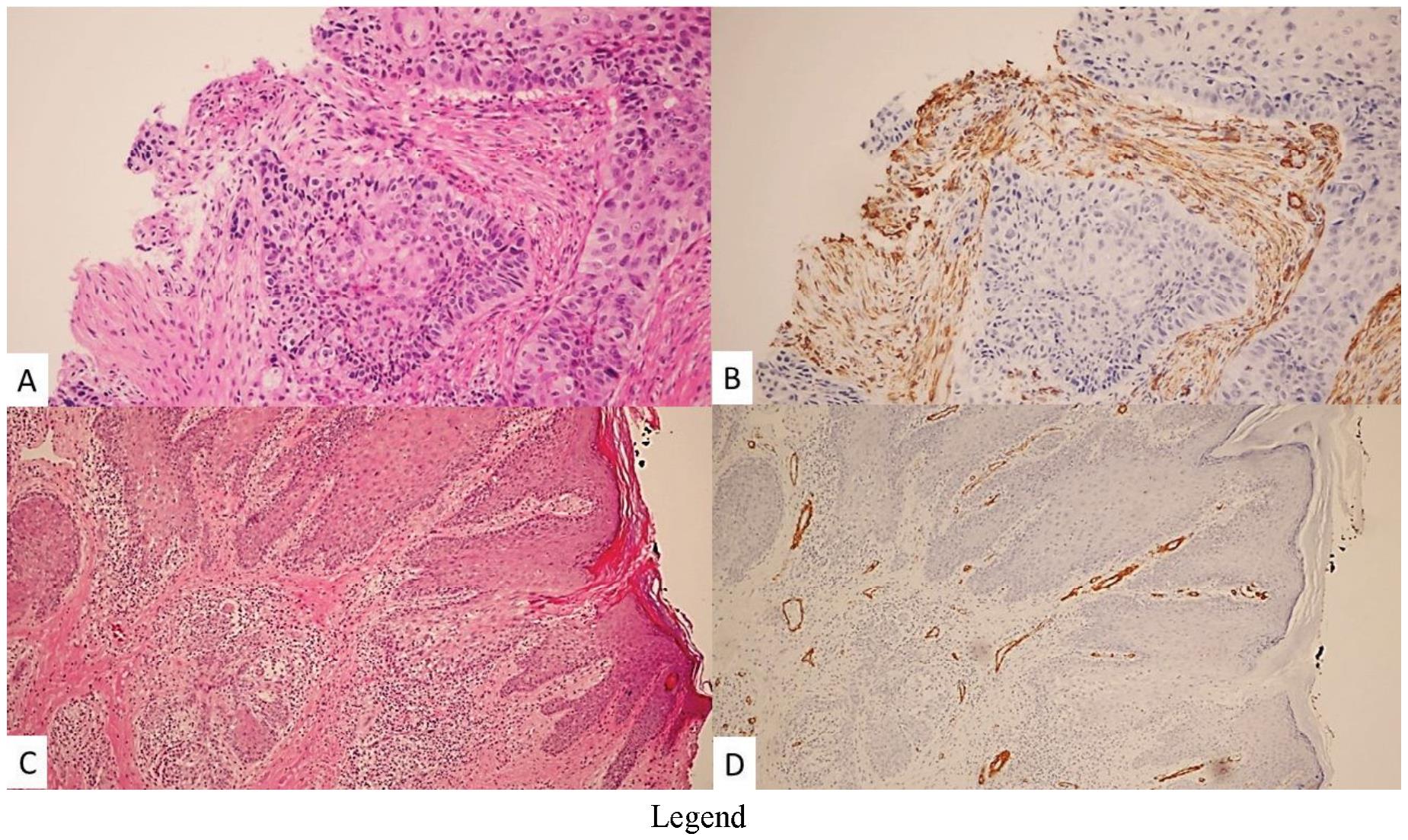
Figure 1 Comparison between SCC (A-B) and PEH (C-D). Alpha smooth muscle actin is strongly expressed in reactive desmoplastic tissue around SCC (B) but negative in PEH (D). Blood vessels’ walls serve as internal positive control (D). (A-B, x200, C-D, xlOO).
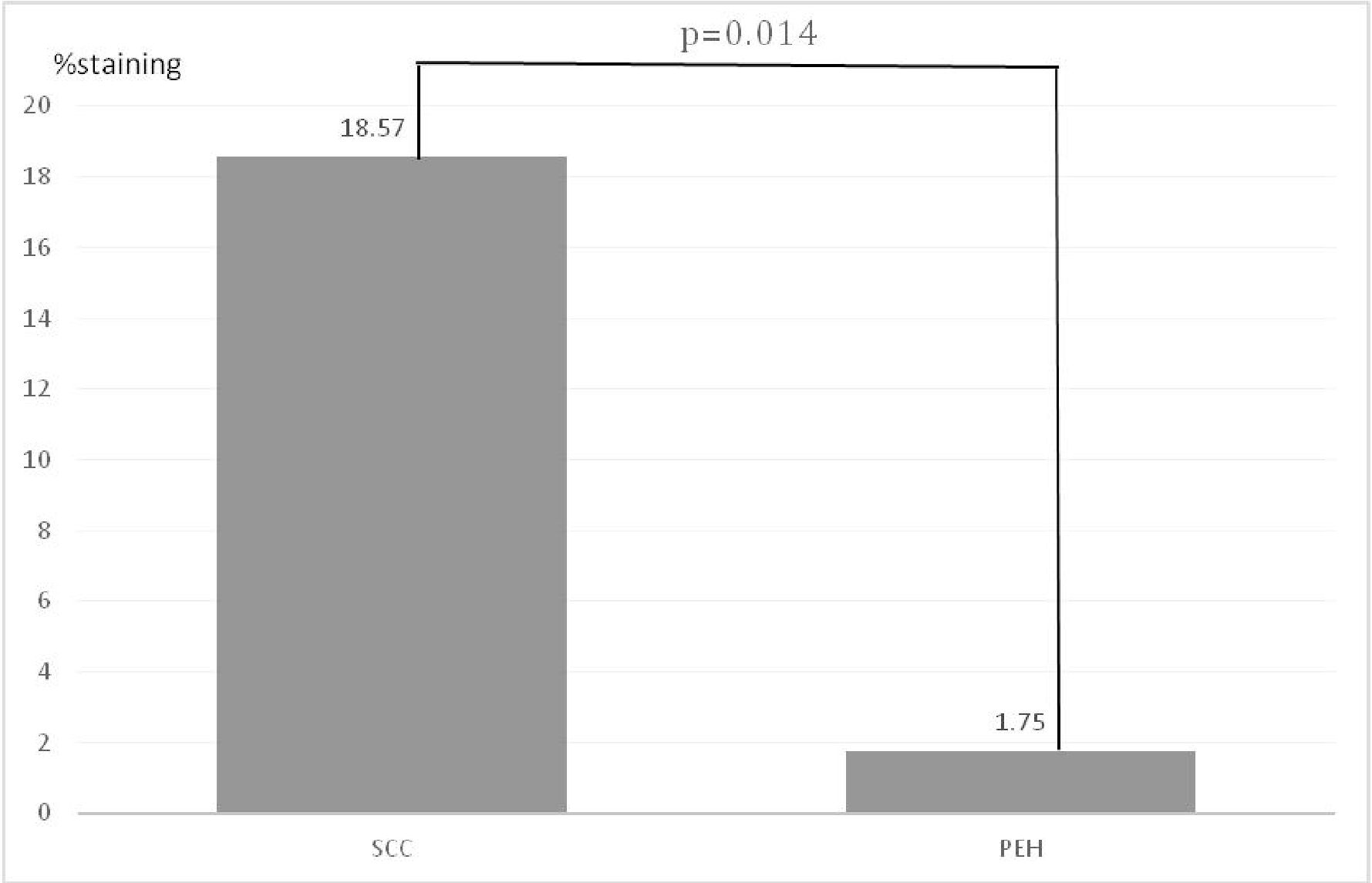
Figure 2 Graph showing positive cases for a-SMA expression with their intensity of expression in both groups
Statistical Analysis
A comparison between groups was carried out by independent student T test. The p-value of less than 0.05 was considered statistically significant. Sensitivity, specificity, positive and negative predictive values at different percentage of staining were calculated.
RESULTS
A wide variation in sites of the lesions was noted in both squamous cell carcinoma and pseu-doepitheliomatous hyperplasia. Details regarding site distribution are summarized in table 1
Squamous cell carcinoma were classified as; well differentiated (7 cases), moderately differentiated (12 cases) and indeterminate (2 cases). Desmoplastic reaction was detected by H&E in 7 out of 21 cases (33.4%).
Two pseudoepitheliomatous hyperplasia had co-existing carcinomas, one of which was well differentiated squamous cell carcinoma and the other of which was verrucous carcinoma. Other related changes included fungal infection (1 case), chronic inflammation (3 cases), organizing inflammation (4 cases) and chronic ulcers (3 cases). Reactive atypia of the epithelium was observed in 1 case.
There were 13 cases of squamous cell carcinoma (61.90%) and 4 cases of pseudoepitheliomatous hyperplasia (20%) whose reactive stroma positive to a-SMA staining. The percentage of staining for reactive stroma of squamous cell carcinoma ranged from 5-90 % (meanl8.57, SD 28.33) while for pseudoepitheliomatous hyperplasia, staining ranged from 5-20 % (mean 1.75, SD 4.67) with the difference between which were statistically significant (p=0.014) (figure 2).
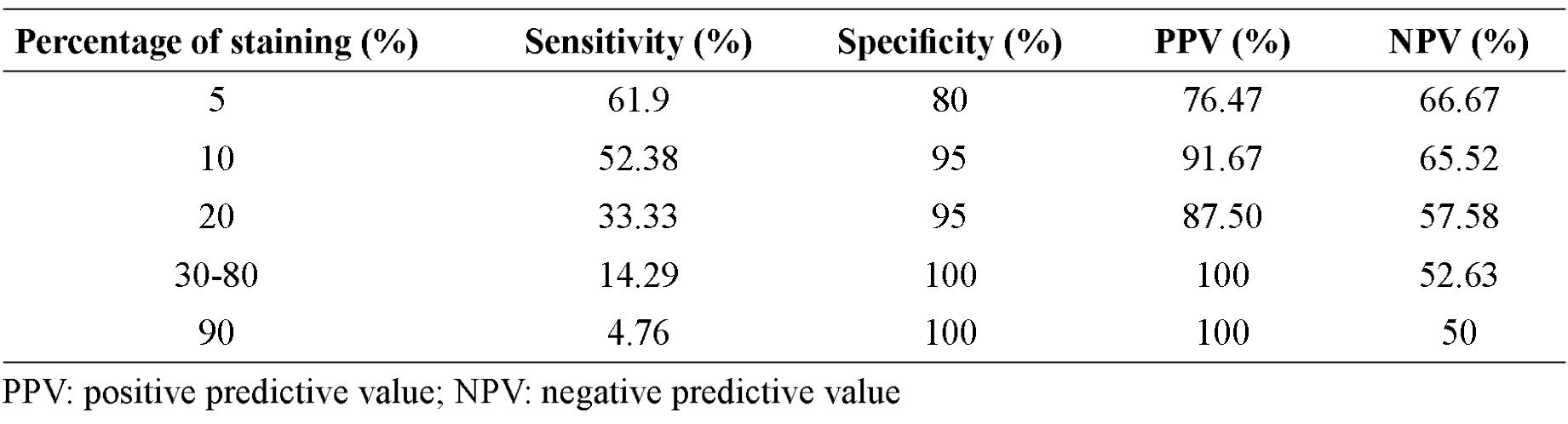
At different cutoff points of staining by which a-SMA was used to differentiate SCC from PEH, the sensitivity was highest at the staining of 5% (61.9%) and the specificity was highest at the staining of 30-90 % (100%). The sensitivity and specificity of various cutoff points of staining are summarized in table 2. From this table, the cutoff value at 10% staining would seem to be the most appropriate option making sensitivity 52.38%, specificity 95%, PPV 91.67%, NPV 65.52 %
Table 1 Site distribution of the cases included in this study
Table 2 Sensitivity, specificity, positive predictive value and negative predictive value in different percentage of a-SMA staining
DISCUSSION
Several studies have shown concordant results on a-SMA overexpression in the stromal reactive tissue around the invasive front of the SCC compared to those adjacent to the normal and benign epithelium2'4. The mechanism of such findings is believed to be due to several cytokines secreted by the tumor cells, one of which is TGF-|i 1 that has ability to induce transdifferentiation of fibroblasts to become myofibroblasts in desmoplastic tissue3-5. This change may even occur at the early stage of cancer development as demonstrated by one study in which high a-SMA expression was found to be present in reactive stroma adjacent to dysplastic epithelium and invasive cancer cells of SCC compared to oral submucosal fibrosis4. Moreover, it has also been observed in a less aggressive cancer like verrucous carcinoma6. This finding is consistently described in several locations eg. in SCC of the skin, cervix, esophagus and upper aerodigestive tract, in ductal carcinoma of the breast and in adenocarcinoma of the pancreas and colon6.
In this study, results are also in concordance with those described in the literature. However, since there has been no study of a-SMA expression on PEH which oftentimes can cause diagnostic problems with SCC, the authors, therefore, included cases of PEH to be studied in parallel with SCC. As anticipated, a-SMA staining was more prominent in reactive stroma of SCC compared to those of PEH. In addition, the authors found that this marker is slightly superior to routine H&E examination regarding sensitivity in detection of myofibroblasts in desmoplastic changes, since, at the cutoff value of 10%, more than 50% of SCC showed expression of this protein whereas, in H&E slides, such changes can be identified by pathologists in only 33.34 % of cases. This, therefore, adds some more value to pathology practice besides H&E examination in detection of invasiveness of cancer cells which theoretically can be used to differentiate SCC from benign lesions.
Despite significant differences between SCC and PEH, there are some limitations in using this marker as a diagnostic tool to distinguish between these two conditions. Firstly, due to a low rate of positivity, this test is not considered a sensitive tool for detection of carcinomas. Secondly, since myofibroblasts are also involved in inflammatory and reparative processes induced by the same cytokines secreted by the invasive cancer (eg. TGF-pt), false positivity may occur in some non-cancerous, inflammatory lesions7 where interpretation should be undertaken with caution. Thirdly, the degree of staining in this series varies widely from case to case (5-90%), causing difficulties for evaluation.
With these limitations, the authors, therefore, do not recommend to rely solely on this marker for definite diagnosis of SCC. Instead, it should be used in combination with other modalities including routine H&E and other immunohistological markers eg. P53, MMP-1 and CD-341-6-8.
Helpful features of H&E slide evaluation for SCC include atypical cell nests with irregular margins embedded in desmoplastic stroma. They usually contain pleomorphic nuclei and frequent mitoses. In contrast, PEH is characterized by irregular cords of epithelial cells downgrowing into the subepithelium with elongated and jagged rete ridges without nuclear atypia or prominent mitoses.
p53 and MMP-1 are usually overexpressed in cancer cells of SCC but the staining is limited at the basal layer of PEH1-8. For CD-34, the staining pattern is reversed to a -SMAthat is its expression is reduced in the reactive stroma adjacent to SCC while for a -SMA, the nearby stroma to cancer cells tend to overexpress this protein3.
In summary, the authors demonstrate that a-SMA can be used as another ancillary tool in addition to H&E, p53, MMP-1 and CD-34, to support SCC diagnosis since it stains more prominently on the reactive stromal tissue of SCC than those adjacent to the proliferative epithelium of the PEH. However, due to some limitations as described earlier in this section, it is not recommended as a definite marker for SCC. Instead,
H&E stain remains the gold standard for definite diagnosis of such lesions. In problematic cases where definite diagnosis cannot be made with the help of all available modalities, re-biopsy should be considered because due to serious morbidities from false positive diagnosis, such error should not be allowed to occur.
REFERENCES
1. Zarovnaya E, Black C. Distinguishing pseu-doepitheliomatous hyperplasia from squamous cell carcinoma in mucosal biopsy specimens from the head and neck. Arch Pathol Lab Med. 2005 Aug;129(8):1032-6.
2. Eliene-Magda de-Assis, Luiz-Gustavo-Garcia-Santos Pimenta, Edson Costa-e-Silva, Paulo-Eduardo-Alencar Souza, Martinho-Campolina-Rebello Horta. Stromal myofibroblasts in oral leukoplakia and oral squamous cell carcinoma. Med Oral Patol Oral Cir Bucal. 2012 Sep l;17(5):e733-8: doi: 10.4317/medoral. 17834.
3. Li Q, Huang W, Zhou X. Expression of CD34, a-Smooth Muscle Actin and Transforming Growth Factor-pi in Squamous Intraepithelial Lesions and Squamous Cell Carcinoma of the Cervix. The Journal of International Medical Research. 2009;37:446-54.
4. Rao KB, Malathi N, Narashiman S, Rajan ST. Evaluation of myofibroblasts by expression of alpha smooth muscle actin: a marker in fibrosis, dysplasia and carcinoma. J Clin Diagn Res. 2014 Apr;8(4):ZC14-7.
5. Sridhara SU, Choudaha N, Kasetty S, Joshi PS, Kallianpur S, Tijare M. Stromal myofibroblasts in nonmetastatic and metastatic oral squamous cell carcinoma: An immunohistochemical study. J Oral Maxillofac Pathol. 2013 May;17(2):190-4.
6. Paral KM, Taxy JB, Lingen MW. CD34 and alpha smooth muscle actin distinguish verrucous hyperplasia from verrucous carcinoma. Oral Sing Oral Med Oral Pathol Oral Radiol. 2014 Apr;117(4):477-82.
7. Micallef L, Vedrenne N, Billet F, Coulomb B, Darby I A, Desmouliere A. The myofibroblast, multiple origins for major roles in normal and pathological tissue repair. Fibrogenesis Tissue Repair. 2012;5(Suppl 1 Proceedings of Fibro-proliferative disorders: from biochemical analysis to targeted therapiesPetro E Petrides and David Brenner) :S5.
8. Choi KH, Lim JH, Lee JH, Kwon KS, Lee H, Park HS, et al. Expression of Claudin-1, p53 and E-cadherin in Pseudoepitheliomatous Hyperplasia and Squamous Cell Carcinoma of the Head and Neck. The Korean Journal of Pathology. 2008;42:287-93.


