Acute and subchronic oral toxicity of xanthones extracted from the pericarp of Garcinia mangostana Linn.in rat
Kanokporn Chayaburakul1, Patamaporn Sukplang2, Thanaporn Pinpart3
and Kanjana Kengkoom4*
1, 2Department of Medical Science, Faculty of Science, Rangsit University, Patumthani 12000
E-mail: kanokporn.c@rsu.ac.th,
3, 4Office of Laboratory Animal Production, National Laboratory Animal Center, Mahidol University, Thailand
Correspondence : Kanjana Kengkoom
E-mail: directac@mahidol.ac. th
Received 30th March 2015; Accepted 23 September 2015
ABSTRACT
The purpose of this research was to study the acute and subchronic toxicity of crude mangosteen pericarp hydro extract, and determine the appropriate and safe amount of extract that may be consumed, through testing with Sprague Dawley rats of both sexes. The soft layer of the inner pericarp mangosteen fruits was immersed in water and boiled at 70 OC, and the supernatant was concentrated and spray-dried into a powder. For the acute toxicity test, 5 rats of each sex were administered intragastrically with the extract powder dissolving in water at a dose of 2000 mg/kg BW. The animals were then observed closely during the first day post-treatment, before being observed continuously for 14 days. The results of the acute toxicity test showed no abnormalities. For the subchronic toxicity examination, 10 male and 10 female rats were placed in each dosage group for 3 months. The extract was administered orally at doses of 10, 50, and 100 mg/kg BW/day; distilled water was given to the control group. The extract induced no significant effect on body weight, hematology or the relative weight of organs (brain, heart, lungs, thymus, liver, spleen, kidneys, adrenal glands, testes, and ovaries). Histological examination of the liver, lungs, spleen and heart revealed normal appearance in most all extract treated rat. Only the kidney of one female rat receiving magosteen pericarp hydro-extract 100 mg/kg BW/day, showed a protruded nucleus in the proximal convoluted tubule. This may be associated with the elevated secretion of creatinine, though creatinine levels remained within reference parameters. Furthermore, the subjects in this group presented torn glomeruli; this may be due to a defect during the histology process, since no white blood cells or inflammatory cells were identified. The results of this experiment indicated that a safe dosage of the mangosteen pericarp hydro-extract would be < 100 mg/kg BW/day. The effects of subchronic kidney exposure to mangosteen pericarp on kidney require further investigation.
Keywords: mangosteen pericarp hydro extract, acute and subchronic toxicity, histological examination, kidney, clinical chemistry
INTROUCTION
The mangosteen (Garcinia mangostana Linn.) is a tropical tree endemic to India, Thailand, Myanmar, Malaysia, the Philippines, and Sri Lanka. Mangosteen fruits are sweet, and are eaten in many cultures. The mangosteen pericarp has been used as a traditional medicinal agent by Southeast Asians for centuries to treat skin infections, wounds1,2 and amebic dysentery3,4. In Ayurvedic medicine, mangosteen pericarp has a wide range of applications, including the treatment of inflammation, diarrhea5, cholera, and dysentery6.
Four prenylated xanthones from mangosteen pericarps; a-mangostin, P-mangostin, y-mangostin. and methoxy-P-mangostin, have been demonstrated to exhibit a variety of biological activities, including anti-inflammatory properties in in-vitro models, C6 rat glioma cells, including rabbit thoracic aorta and guinea pig trachea, and several models in vivo in rats7,8.
They also possess antioxidant properties in 2,2 diphenyl-1-picrylhydrazyl (DPPH) and radical scavenging activity assays9,10,11,12, as well as the ferric thiocyanate method9. In addition they are also studies that demonstrated their anti-bacterial potential13 and effect on cancers, such as hepatocellular carcinoma, SKBR3 human breast cancer14, human leukemia15, and various human cancer cells16,17.
Previous research has focused on the potential benefits of mangosteen-derived xanthones, and some toxicity tests have also been published18,19,20. However, the Food and Drug Administration of Thailand has not permitted their sale in the Thai market. This restriction may be result from the present available data that only ethanolic extract was tested. Thus in this study, water was used as the solvent in preparation of extract for conducting both acute toxicity and subchronic toxicity testing for mangosteen pericarp.
MATERIALS AND METHODS
Plant material and preparation of mangosteen pericarp extract
Mangosteen (G. mangostana) fruits were purchased in Chantaburi Province, Thailand. The fresh pericarps were washed, and the inner soft part (purple color) was isolated for extract preparation. One kilogram of inner soft fresh mangosteen pericarp was heated with 1 liter of distilled water at 70 °C for 2 hr; the supernatant was then concentrated by evaporation under pressure for about 6-8 hr. The concentrated extract solution was then heated at 65 °C for 30 min. The concentrated extract was f urther evaporated with a rotary evaporator at 55 °C under reduced pressure, then placed in a spray drying machine. The yield of dried mangosteen pericarp was about 0.67% (w/w). The content of a-xanthone, a major biologically active compound in mangosteen pericarp extract, was about 21.23% by HPLC analysis. The dried extract was stored until use at 4 °C in sterilized, sealed plastic containers away from light. The same batch of extract was used throughout this study.
Acute and subchronic oral toxicity study
A total of 100 specific-pathogen-free bred Sprague Dawley (SD) rats (50 male and 50 female rats, weighing about 200-250 g) were selected, 10 rats (5 rats per sex) for the acute test, and 90 rats (45 per sex) for the subchronic toxicity test. Five additional rats were used as sentinel animals to indicate the environmental status in the long-term study. All experiments were reviewed and approved by the National Laboratory Animal Center Animal Care and Use Committee, Mahidol University, Thailand. The animals were housed in a hygienic conventional animal room within the National Laboratory Animal Center, Mahidol University. The room was maintained at 25+1 OC, 55+15% relative humidity and a 12 hour light-dark cycle.
In the acute test (OECD 420 and 408), a maximum test dosage of 2000 mg/kg BW was administered orally as the initial dose level, to 5 fasted female and fasted male rats. They were then observed individually at 0.5, 4, 8, 12, and 24 hours post-dosing, and at least once daily for the next 14 days18192021. In the subchronic-toxicity test (OECD 420 and 408), 90 rats were randomly divided into 4 groups of 20 animals (80 rats; 10 males and 10 females per group). Three of these groups were experimental groups, with 1 control group. Two additional groups (5 rats per group) were used; one was a satellite group for observations of potential reversibility or the persistence of any toxic effects; the other was a sentinel group to monitor the quality of animal management throughout the study. Each animal in treatment groups was administered orally with water-extract of mangosteen pericarp daily, at daily doses of 10, 50, 100, and 0 mg extract/kg BW in experimental groups 1, 2, and 3, and the control group. Rat in the additional group (the satellite group) was fed with100 mg/kg/day of extract orally for 90 days, and then raised without treatment for an additional of 14 days, to assess the reversibility of any adverse effects produced by the highest dose of mangosteen pericarp extract. The last group (sentinel group: 5 animals) was monitored for 14 days longer than the satellite group, to check the quality of animal management throughout the experiment.22 The control group was administered only distilled water without mangosteen pericarp extract. All animals were carefully observed individually for signs of toxicity each day. In both studies, the weights of the individual animals and feed consumption were measured daily.
Mortality and signs of toxicity, hematological and blood clinical chemistry
The animals were observed closely for toxic signs and symptoms, neurobehavioral signs, and death. During the experiment, the weight of each individual rat was determined and recorded weekly. At the end of the treatment period, the animals were fasted overnight, then anesthetized by CO2 inhalation. Blood samples were collected from the posterior vena cava, to determine hematological and serum clinical chemistry values.
Relative organ weights and histopathology
All rats were subjected to gross necropsy. All gross pathological changes were examined microscopically. In the subchronic toxicity test, the brain, heart, spleen, kidneys, liver, lungs, thymus, adrenal glands, testes and ovaries, were removed and weighed separately, to identify relative organ weights. Then, all organs were fixed in 10% neutral buffer formalin (NBF). Fixed specimens were embedded in paraffin wax. Various cross-sections (4 pm) were mounted on glass slides for staining with hematoxylin and eosin (H&E).
Statistical analysis
Parametric and nonparametric statistical analyses were used to analyze the study data. In the parametric analysis, normality was tested using the Kolmogorov-Smirnov test, while homogeneity of variances was tested by one-way ANOVA. The treatment and control groups were compared by student t-test. Differences between groups were considered significant at p<0.05.
RESULTS
Survival and signs of toxicity in the acute toxicity tests
No treatment-related mortality or signs of toxicity were observed in either acute or subchronic experiments. Treatment and concurrent control groups demonstrated similar manifestations.
Body weights in the acute toxicity tests
All Sprague Dawley rats fed with water-extracted mangosteen pericarp by intragastric gavage survived the 14-day experiment. They did not exhibit any signs of toxicity up to the end of the experimental period. There were no obvious differences in body weight, growth rate, internal relative organ weights or blood chemistry between the treatment and control groups. Histological examinations of the internal organs found no pathological abnormalities.
Body weights in the subchronic toxicity tests
Overall, the average body weights (g) and the growth of the 4 groups of Sprague Dawley rats fed orally with mangosteen pericarp extract for the subchronic toxicity test gradually increased during the testing period. Treatment induced no mortality in either gender; however, slight decreases in body weight among the female rats treated with 50 mg/kg BW/day of mangosteen pericarp extract, between days 79-86, were noted; this was non-significant when compared with the control group (Figure 2). The average body weights (g) and the growth of the 4 groups of male rats did not change significantly, when compared with the control group (Figure 1).
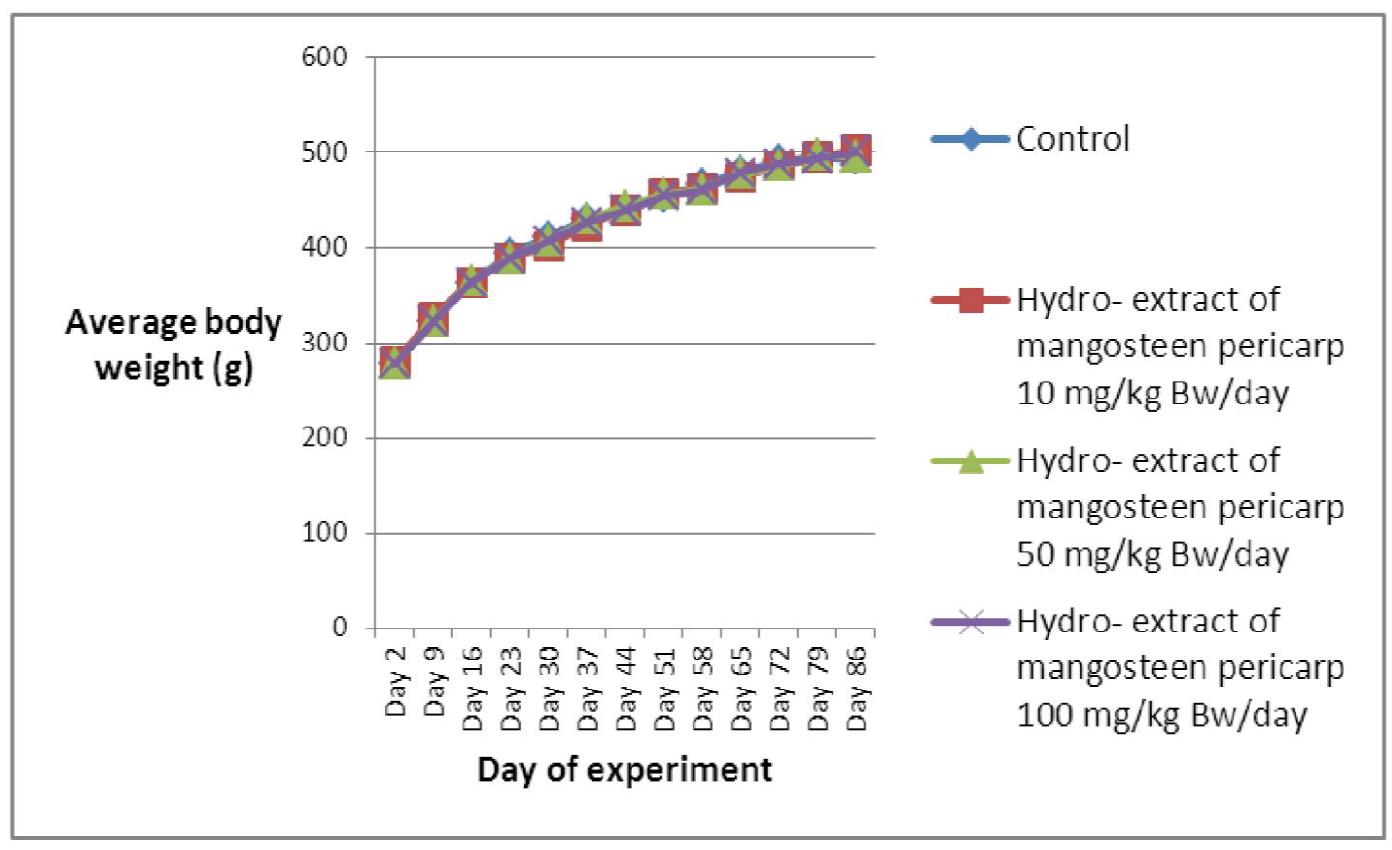
Figure 1 Average body weights (g) and growth of 4 groups of 10 male Sprague Dawley rats; 3 experimental groups fed orally with mangosteen pericarp extract at dose of 5, 10, and 100 mg/kg BW/day for subchronic toxicity testing at and 1 control group fed with distilled water.
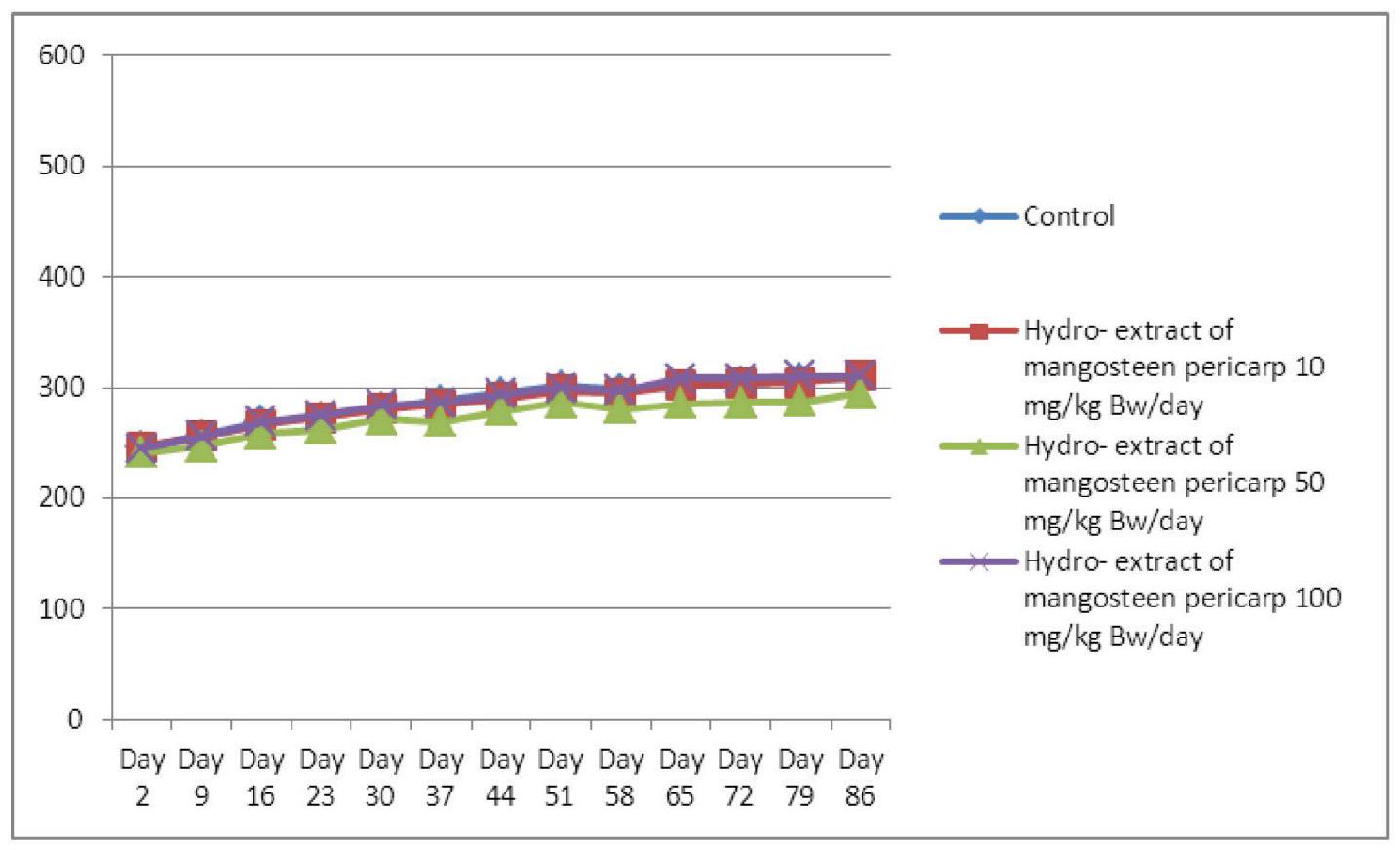
Figure 2 Average body weights (g) and growth of 4 groups of 10 female Sprague Dawley rats, comprising 3 experimental groups fed orally with mangosteen pericarp extract at doses of 10, 50, and 100 mg/kg BW/day for subchronic toxicity testing and 1 control group fed with distilled water
Table 1 Average relative organ weights (g)/100 g BW of the 4 groups of 20 Sprague Dawley rats (10 males and 10 females per group), in subchronic toxicity test. Three experimental groups were treated with mangosteen pericarp extract at dose of 10, 50, and 100 mg/kg BW/day. The control group was fed orally with distilled water. The data were presented as mean±SD.
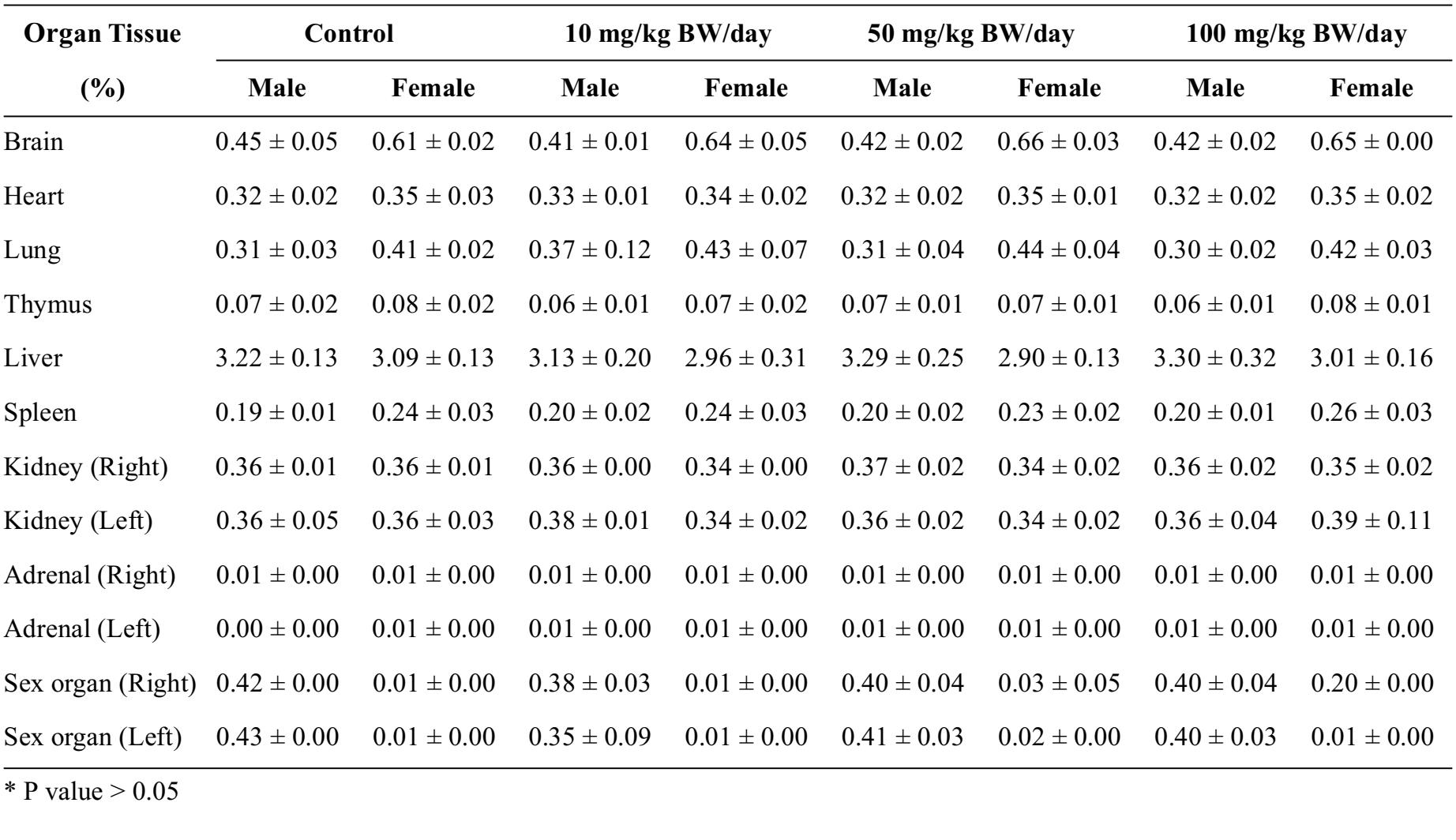
Table 2 Hematology values for the 4 groups of 20 male and female Sprague Dawley rats. The 3 experimental groups received 10, 50, or 100 mg/kg BW/day of mangosteen pericarp extract. The control group was administered distilled water without pericarp extract. The data were presented as mean±SD. Hematology Control Extract10 mg/kg BW/day Extract 50 mg/kg BW/day Extract100 mg/kg BW/day
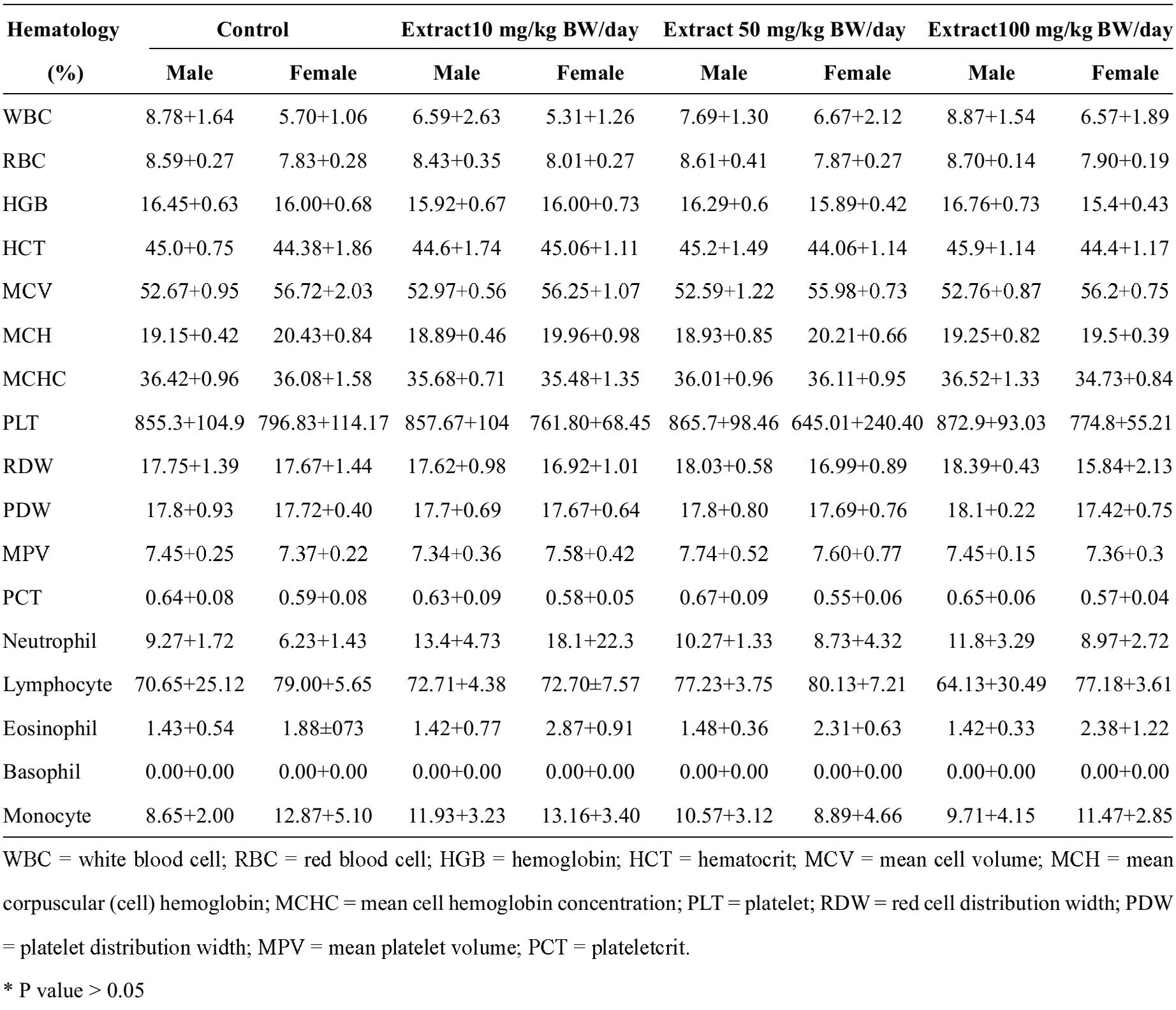
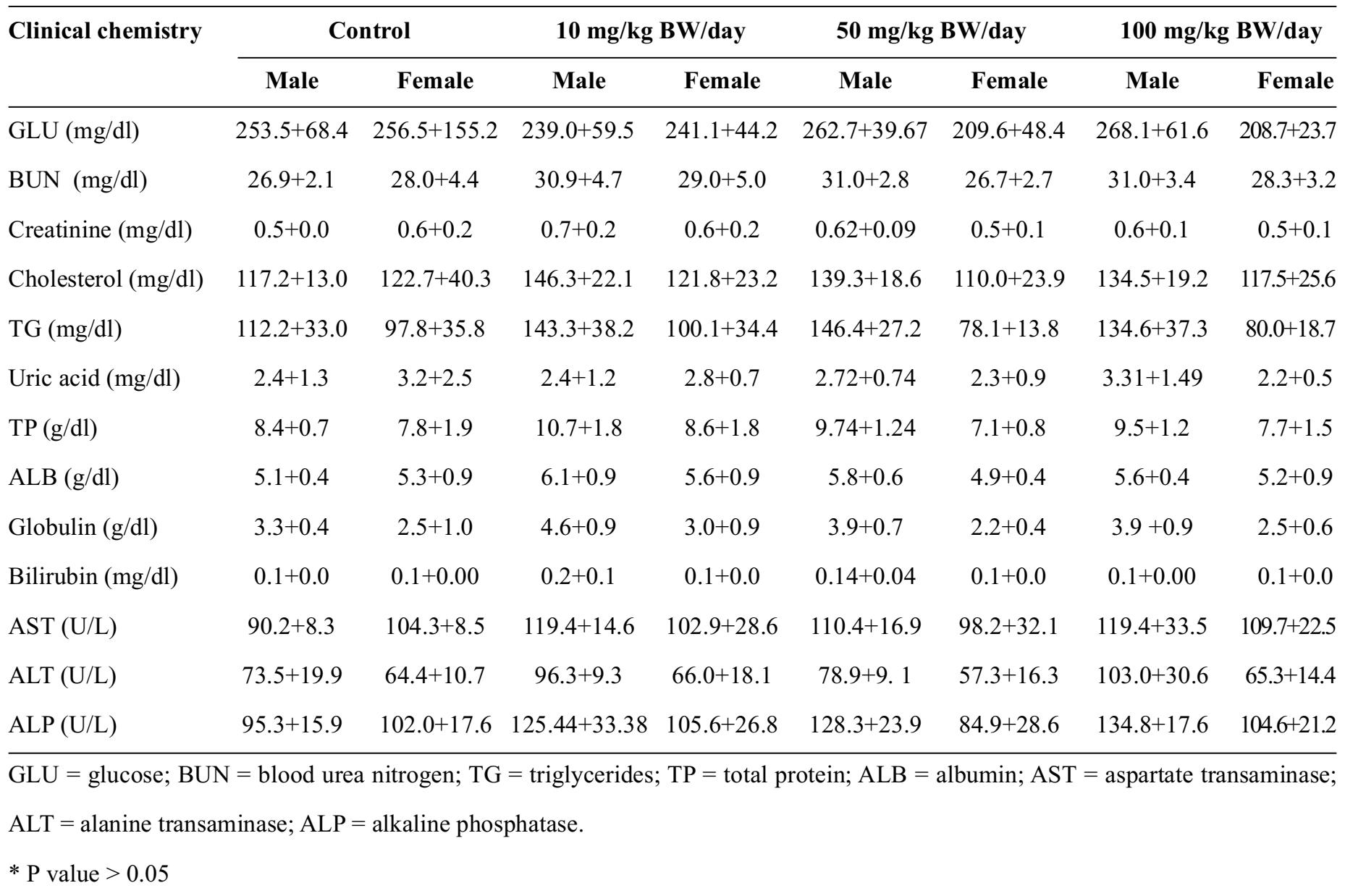
Table 3 Clinical blood chemistry values for the 4 groups of 20 male and female Sprague Dawley rats (10 of each gender per group) for subchronic toxicity test, The control group was fed orally with distilled water, and three experimental groups were administered mangosteen pericarp extract for subchronic toxicity test at doses of 0, 10, 50, 100 mg/kg BW/day.
Organ weights
The brain weights of the male rats decreased slightly, while the brains of the females increased slightly in the experimental groups; this change was non-significant (Table 1). The lung weights of the male rats increased slightly in the groups administered mangosteen pericarp extract at 10 and 50 mg/kg BW; the lung weights of the female rats increased slightly in all 3 groups. The lung weights of the male rats in the 10 mg/kg BW group and the females in the 50 mg/kg BW group showed the greatest increase among the 3 groups, though this result was still non-significant compared with the control group (Table 1). The liver weights of the female rats decreased in all 3 groups, but the liver weights of the male rats increased in the 50 and 100 mg/kg BW groups. This result was again non-significant compared with the control group (Table 1), as were the results for all other organs.
Effect of water-extracted mangosteen pericarp on hematological values in the subchronic toxicity tests
The hematological values were examined in all groups of male and female Sprague Dawley rats. The hematological values of the experimental male and female rats were not significantly different from the control group. However, the mean WBC of the female rats in the 50 and 100 mg/kg/day groups increased slightly (6.67+2.12 and 6.57+1.89), compared with the control group, but both of these results were not statistically significant.
The mean platelet levels of the male rats in the groups receiving 50 and 100 mg/kg BW/day increased slightly, while a small decrease was noted for the same test among all groups of female rats. These results were statistically insignificant. The mean monocyte counts increased in all groups of male rats compared with the control group, but not significantly. The mean monocyte counts in the female rats increased slightly only in the 10 mg/kg/day group, and decreased in the groups receiving 50 and 100 mg/kg BW/day when compared with the control group; all differences were insignificant.
Effects of water-extracted mangosteen pericarp on blood clinical chemistry values
Blood clinical chemistry values were measured and analyzed in all groups of male and female Sprague Dawley rats. The levels of creatinine, total protein, globulin, bilirubin, and albumin in the group receiving 10 mg/kg BW were significantly higher than the control group. The alkaline phosphatase levels in the groups receiving 50 and 100 mg/kg BW were significantly higher than the control group (fed only distilled water).
Histopathology in the subchronic toxicity tests
Histological sections (H & E stain) of various organs of the Sprague Dawley rats receiving mangosteen pericarp extract at doses of 0, 10, 50, and 100 mg/kg/day were taken and examined.
All cardiac muscle cells obtained from rats belonging to the 10, 50, and 100 mg/kg/day groups were found to be similar to the control group (Figure 3A-D). Likewise, the photomicrographs of the liver and spleen isolated from these treated rats were also normal in appearance, as in the control group (Figure 3E-F, 4G-H).
As illustrate in the kidney photomicrograph, kidney from the 100 mg/kg/day treated-group shows pathologically damaged. The parietal capsules of some glomeruli are torn (Figure 4B, 4D). But, the blood cells within the glomerular capillaries appear to be normal red blood cells instead of white blood cells or inflammatory cells (Figure 4F).
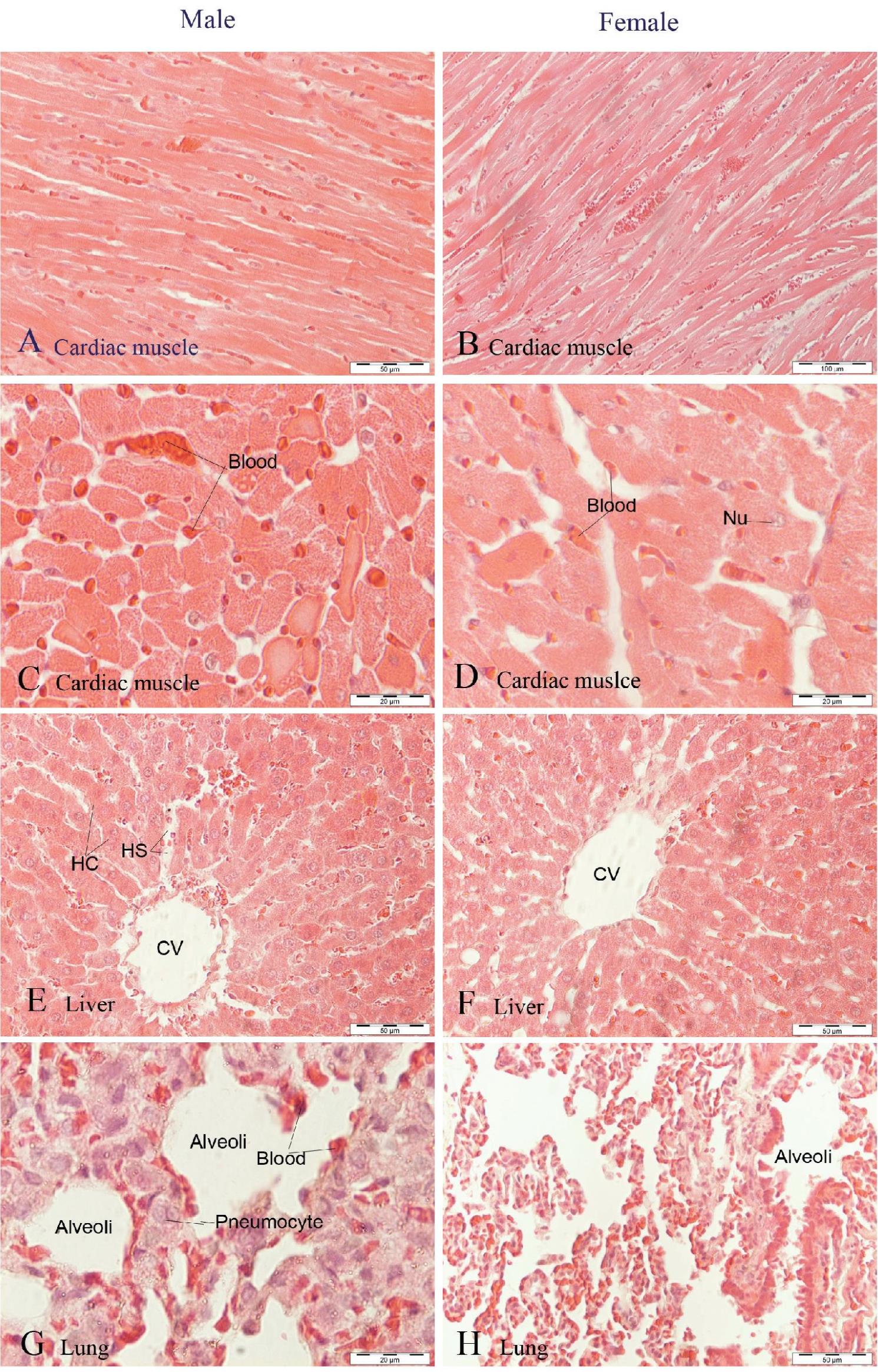
Figure 3 Photomicrographs of H&E staining of tissues obtained from male and female rats after chronic administration of water-extracted mangosteen pericarp (100 mg/kg BW) for 3 months; longitudinal section of cardiac muscle or myocardial cells is illustrated in A and B, cross-section of cardiac muscle cell is shown in AC and D, hepatocyte cells, hepatic cord (HC), hepatic sinusoid (HS), and the centralvein (CV) are presented in E and F, pneumocyte cells of the lung and alveoli are illustrated in G and H.
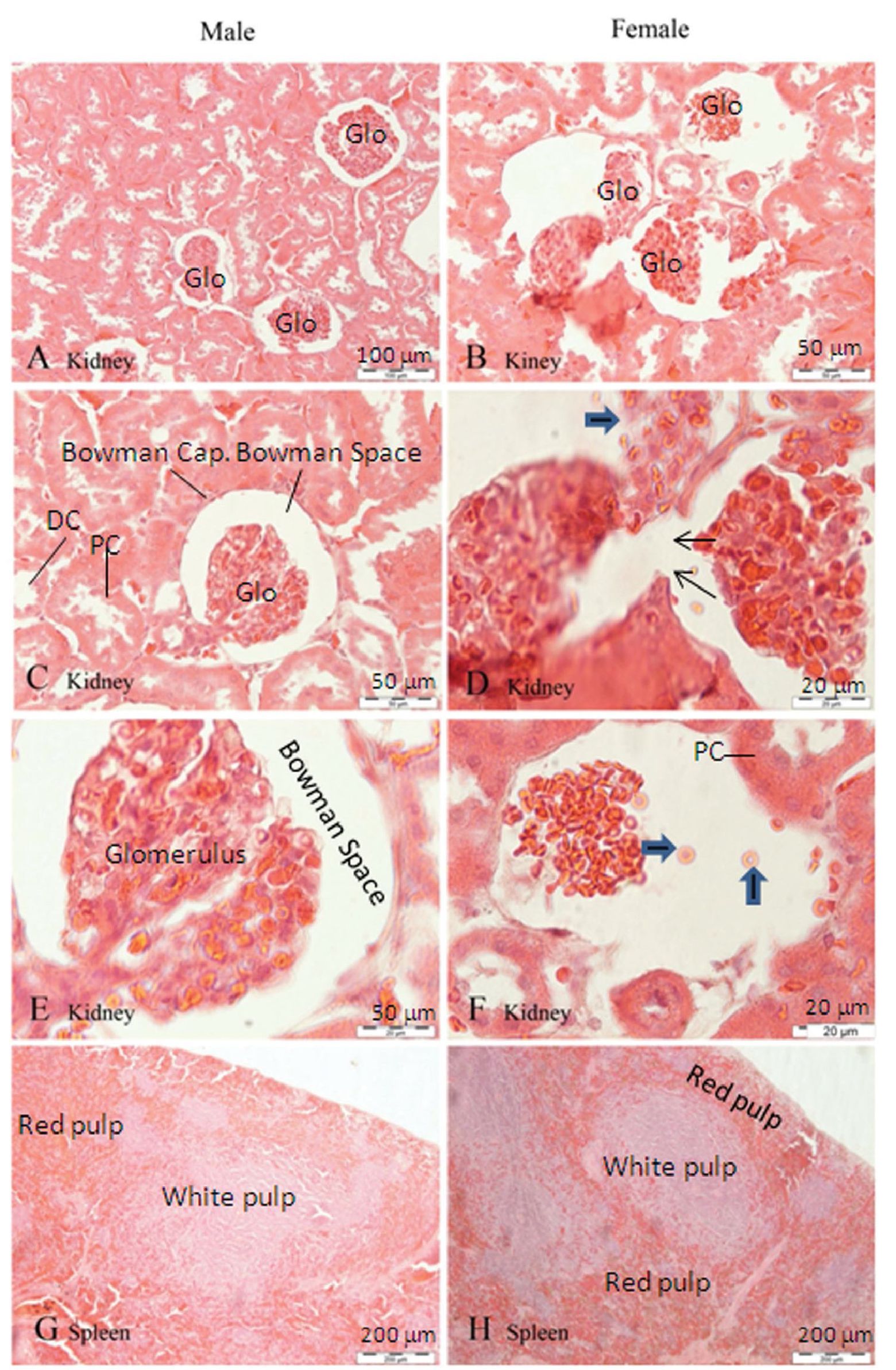
Figure 4 Photomicrographs of H&E staining of tissues obtained from male and female rats after chronic administration of water extracted mangosteen pericarp (100 mg/kg) for 3 months. Kidney is shown in A to F and spleen in G-H. Kidney consists of glomerulus (Glo), Bowman’s capsule and Bowman’s space, proximal convoluted tubule (PC), and distal convoluted tubule (DC). Figure B illustrates the tom glomerulus and Bowman’s capsule observed in a female kidney. Figure D shows the magnified torn Bowman capsule (thin arrow) and red blood cell leakage into the Bowman’s space (thick arrow) in figure F.
DISCUSSIONS
The mangosteen pericarp extract used in this study differs from that in other experiments. This study used water as a solvent instead of ethanol, and prepared the concentrated extract from only the soft inner part of the mangosteen pericarp, due to its higher concentrations of xanthone. Several previous studies have illustrated the numerous medicinal properties of xanthones. Thus, this study extracted xanthones from the mangosteen pericarp while minimizing the amount of tannins, which can cause weight loss18192021.
The oral acute toxicity test result in this study showed no lethality, abnormal behavior, changes in body weight or hepatopathological effects on the animals, as also reported by Hutadilok-Towatana20 and Priya20.
Peaslee and Einhellig22 found that mice placed on a diet containing elevated levels of tannic acid showed retarded growth rates, compared with those administered low-tannin diets. This result concurs with that of the study by Chivapat22. which used 95% ethanolic extract of mangosteen pericarp. However, Hutadilok-Towatana20 found that 3-month oral administration of 50% ethanol extract in Wistar rats did not affect body weight at any time-point. From these previous studies, it can be assumed that the use of lower percentages of ethanol to obtain mangosteen pericarp extract results in lower levels of tannin in the extract; tannin has been shown to affect body weight negatively. Thus, in this experiment, the mangosteen pericarp was extracted using water after the outer hard shell had been removed, so reducing tannin levels; this resulted in no significant weight loss in this experiment. This study also used 100 mg/kg BW/ day as the highest experimental dosage, instead of 500 mg/kg BW/day as used by Chivapat21. The result obtained indicated is experiment showed no mortality and no signs or symptoms of toxicity, as observed by Hutadilok-Towatana19 and Jujun23.
The relative weights of the various organs in this study were not significantly different across the experimental groups. The results for the brain, lungs, and liver did show some variation, though this is likely due to normal biological variations among rats, rather than the effect of the pericarp extract.
Hematological analysis of the blood samples from the male Sprague-Dawley rats revealed no significant difference from the control group, confirming the results of Jujun24 who studied the same breed of rat.
The rise in creatinine levels among the male rats receiving 10 mg/kg BW/day might not be due to kidney function (glomerular filtration rate and/ or proximal convoluted tubule secretion), because the mean creatinine level remained within the normal range (0.51-0.66 mg/dl) according to normative data from the National Laboratory Animal Center, Mahidol University. Histology of the male kidney receiving 10 mg/kg BW/day was normal in appearance.
An increase in bilirubin and alkaline phosphatase could affect liver function. Nevertheless, the significantly higher levels of total protein, globulin, and albumin, do not reflect abnormal liver function. Histological examination of the liver illustrated normal appearance, so the increase in bilirubin and alkaline phosphatase levels was not interpreted as a dysfunctional rat liver.
The rise in total protein, globulin, and albumin, might be caused by the dehydration of this group of male rats. It could not be attributed to the mangosteen pericarp extract, since this trend was not dose-dependent, and there were no clinical signs or abnormal liver histopathology. This change is considered incidental, and thought to be congenital and a common genetic trait in many strains of rat24.
Histopathological examination of the lungs of male and female rats in the highest-dose group revealed decreased incidence of bronchiole and alveolar development (large pneumocyte). This may not be due to the extract, since all groups showed the same characteristics as the control group.
Histopathology of female rat kidneys in the highest-dose group revealed torn parietal capsules and glomeruli, and numerous exposed red blood cells, but no white blood cells were observed, implying that no inflammation had occurred. This means the damage is likely to have occurred due to an accident during the histology process. This finding is therefore not attributable to the mango-steen-pericarp extract. The protrusion of nuclei in the proximal convoluted tubule is considered a consequence of the increasing levels of creatinine secreted in this area. Thus, it is possible that receiving the highest doses of xanthones for 3 months might affect proximal convoluted tubule function, even if creatinine elevation remains within the reference range. This finding requires further study. Other histopathological work in the other treatment groups detected neither dose dependency nor significant difference; therefore, it can be concluded that this daage was not due to the mangosteen pericarp extract.
CONCLUSIONS
The acute toxicity test and the 3-month oral administration of water-extracted mangosteen pericarp to Sprague Dawley rats at doses of 10, 50, and 100 mg/kg BW/day did not produce any phar-macotoxic signs or abnormal hematological values. Water-extracted mangosteen pericarp, at doses of 100 mg/kg BW/day, may slightly affect the kidneys. While this finding requires further study, there was no observable effect on liver, heart, or lung function. This study concludes that long-term usage of water-extracted mangosteen pericarp, at doses < 100 mg/ kg BW/day, had no adverse health effects on Sprague Dawley rats.
ACKNOWLEDGEMENCES
This research was funded by Research Institute of Rangsit University, Rangsit University and partial supported by the National Laboratory Animal Center, Mahidol University.
REFERENCES
1. Mahabusarakam, W., Wiriyachtra, P., & Taylor, W. Chemical constituents of Garcinia mango-stana. J Nat Prod 1987; 50: 474-478.
2. Pierce S.C. A Thai Herbal Scotland: Findhorn Press. 2003; 118
3. Garnett M., & Sturton S. D. Garcinia mango-stana in the treatment of amoebic dysentery. Chin Med J, XLVt f932: 969-973.
4. Chopra, R. N., Nayar, S. L, & Chopra, I. C. Glossary of Indian Medicinal Plants. The National Institute of Science Communication and Information Resources. New Delhi: India 1956: 123.
5. Balasubramanian, K., & Rajagopalan, K. Novel xanthones from Garcinia mangostana, structures of BR-xanthone-A and BR-xanthone-B. Phytochemistry1988; 27: 1552-1554.
6. Sen, A. K., Uusvuori, R., & Hase, T. A. A xan-thone from Garcinia mangostana. Phytochemistry 1980; 19: 2223-2225.
7. Nakatani, K., & Yamakuni, T. Gamma-man-gostin inhibits inhibitor-kappaB kinase activity and decreases lipopolysaccharide-induced cyclooxygenase-2 gene expression in C6 rat glioma cells. Mol Pharmacol 2004; 66: 667-674.
8. Chairungsrilerd, N., Furukawa K., Ohta T., Nozoe, S., & Ohizumi, Y. Histaminergic and serotonergic receptor blocking substances from the medicinal plant Garcinia mangostana. Planta Med 1996; 62: 471-472.
9. Yoshikawa M., Harada E., Miki A., Tsukamoto, K., Liang. S. Q., Yamahara, J., & Murakami, N. Antioxidant constituents from the fruit hulls of mangosteen (Garcinia mangostana L.) originating in Vietnam. Yakugaku Zasshi 1994; 114: 129-133.
10. Leong, L., & Shui, G. An investigation of antioxidant capacity of fruits in Singapore markets. Food Chem 2002; 76: 69-75.
11. Weecharangsan, W., Opanasopit, P., Sukma M., Ngawhirunpat, T., Sotanaphun, U., & Sirpong, P. Antioxidative and neuroprotective activities of extracts from the fruit hull of mangosteen (Garcinia mangostana Linn.). Med Princ Pract 2006; 15: 281-287.
12. Chomnawang, M. T., Surassmo, S., Nukool-kam, V. S., & Gritsanapan, W. Effect of Garcinia mangostana on inflammation caused by Propionibacterium acnes. Fitoterapia 2007; 78: 401-408.
13. Chomnawang, M. T., Sakagami, S. S., Nukool-karn, V. S., & Gritsanapan, W. Antimicrobial effects of Thai medicinal plants against acne-inducing bacteria. J Ethnopharmacol 2005; 101: 330-333.
14. Ho, C. K., Huang, Y. L., & Chen, C. C. Garci-none E, a xanthone derivative, has potent cytotoxic effect against the hepatocellular carcinoma cell lines. Planta Med 2002; 68: 975-979.
15. Moongkarndi, P., Kosem, N., Kaslungka, S., Luanratana O., Pongpan, N., & Neungton, N. Antiproliferation, antioxidation and induction of apoptosis by Garcinia mangostana (mangosteen) on SKBR3 human breast cancer cell line. J Ethnopharmacol 2004; 90:161-166.
16. Matsumoto, K., Akao, Y., Kobayashi, E. Oh-guchi, K., Ito, T., Tanaka, T., Linuma, M., & Nozawa, Y. Induction of apoptosis by xanthones from mangosteen in human leukemia cell lines. J Nat Prod 2003; 66: 1124-1127.
17. Watjen, W„ Weber, N„ Lou, Y. J.. Wang, Z. Q., Chovulou, Y., Kampkotter, A., Kahl, R., & Proksch, P. Prenylation enhances cytotoxicity of apigenin and liquiritigenin in rat H4IIE hepatoma and C6 glioma cells. Food Chem Toxicol 2007; 1: 119-124.
18. Bae, E. Y., Na, M., Njamen, D., Mbafor, J. T., Fomum, Z. T., Cui, L., Choung, D. H., Kim, B. Y., Oh, W. K., & Ahn, J. S. Inhibition of protein tyrosine phosphatase IB by prenylated isoflavo-noids isolated from the stem bark of Erythrina addisoniae. Planta Med 2006; 10: 945-948.
19. Hutadilok-Towatana, N., Reanmongkol, W., Wattanapiromsakul, C., & Bunkrongcheap, R. Acute and subchronic toxicity evaluation of the hydroethanolic extract of mangosteen pericarp. J Med Plants Res 2010; 4: 969-974.
20. Priya, V. V., Jainu, M., Mohan, S. K., & Karthik, B. Toxicity study of Garcinia mangosteen Linn. Pericarp extract in rats. Asian J Exp Biol Sci 2010; 1: 633-637.
21. Chivapat, S., Chavalittumrong, P., Wong-sinkongman, P. Phisalpong, C., & Rungsipipat, A. Chronic toxicity study of Garcinia mango-stana Linn, pericarp extract. Thai J Vet Med 2011; 41: 45-53.
22. Peaslee, & Einhellig. Tannic acid-induced alterations in mouse growth and pituitary melanocyte-stimulating hormone activity. Toxicol Appl Phramacol 1973; 25: 507-514.
23. Jujun, P., Pootkham, K., Pongpaibul, Y., Duan-grat, C., & Tharavichitkul, P. Acute and repeated dose 28-day oral toxicity study of Garcinia mangostana Linn. rind extract. CMU J Nat Sci 2008;7: 199-208.
24. King, W. W., & Russel, S. P. Metabolic, traumatic, and miscellaneous disease. In: The Laboratory Rat. M.A. Sucklow, S.H. Weibroth and C.L. Franklin (eds) Burlington: Elsevier AC 2006.


